Figure 67-1 Patient with episode of erythromelalgia. Note bright red feet that have a mottled, blue or purple appearance between episodes. Also note the flushed and red appearance of ears and face.
Based on the history of painful flushing, a diagnosis of EM was made. She was treated unsuccessfully with a variety of regimens for the EM.
Her examination was significant for marked redness and allodynia of the distal limbs, face, nose, and ears. Between episodes of EM, the feet and hands were cyanotic and purple in appearance. Investigations demonstrated changes consistent with EM: noninvasive arterial studies demonstrated increased local temperature and blood flow in the extremities during symptoms. Neurologic studies revealed changes suggestive of small fiber dysfunction: quantitative sensory testing revealed hyperalgesia (Table 67-1), thermoregulatory sweat test (TST) showed marked anhidrosis (95% of body surface) (Fig. 67-2). A skin biopsy showed nonspecific changes, with normal eccrine glands.
Table 67-1 Quantitative sensory test results, showing elevation of vibration threshold, normal threshold to cold and hypersensitivity to heat-pain
| Vibratory threshold | |
| Percentile: >99 | |
| Cooling threshold | |
| Percentile: 40 | |
| Heat-pain | |
| HP: 5.0 | Percentile: 60 |
| HP: 0.5 | Percentile: 0 |
| HP: 5.0–0.5 | Percentile: 1 |
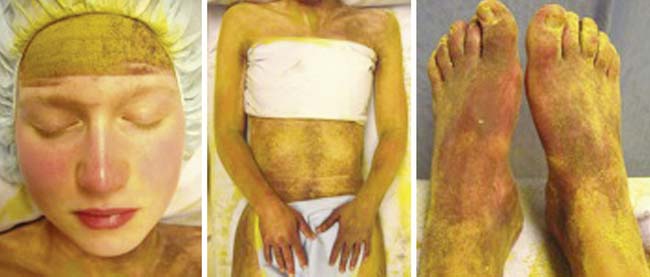
Figure 67-2 TST images showing the marked anhidrosis. Therefore, the indicator powder is yellow-orange when dry and becomes purple when wet. A normal sweating subject will be completely purple at the end of the test: the face is not tested. Our patient was almost completely anhidrotic: the areas that appear red-purple in the images are actually due to her condition, not to sweat. She had only very light, patchy sweating over the trunk and the forehead.
CONCLUSION
The case is instructive because it depicts how the painful flushing of EM can be associated with profound disability. The physical findings of flushing during symptoms and a cyanotic appearance between symptoms were characteristic. The findings on investigation substantiated recent observations from our group and others of increased local blood flow and local increased temperatures during episodes of EM, and of an underlying small fiber neuropathy.
Most cases of EM are idiopathic, but the history of preceding infectious mononucleosis in this patient is interesting: it is conceivable that this was a trigger for the onset of the EM in this patient. Forms of EM secondary to underlying diseases, particularly myeloproliferative diseases, have been described.1
EM is a clinical syndrome describing painful flushing of the extremities, not a specific disorder. Small fiber neuropathies are strongly associated with EM as demonstrated by physiologic studies, particularly the TST and quantitative sudomotor axon reflex test (QSART), with up to 88% of EM patients demonstrating abnormalities on these tests, even if there was no direct correlation between the areas of altered sweating and the symptomatic affected extremities. This latter result should not be surprising because somatic and autonomic fibers may be selectively affected in different small fiber neuropathies.2,3
Skin biopsies in cryptogenic forms of EM are non specific. The most common findings include endothelial swelling, capillary hyperplasia and ectasia. In a small study looking at epidermal nerve fiber density we found a relative decrease in 13 of 16 biopsies, and capillary ectasia was noted.4 Overall, the results were not overwhelming, again in keeping with the complexity and multifactorial pathogenetic causes of this syndrome.4
Inherited forms have been reported. A specific mutation of the sodium channel Nav 1.7 has been described in studies of a number of families in whom EM was familial.5 We did not find this mutation to be present in patients tested at our institution, both with familial or sporadic (the majority) forms of EM.
From a management standpoint, treatment for EM has paralleled the management of neuropathic pain in many ways. Treatment with topical agents (such as the lidocaine patch or a combination gel of amitriptyline and ketamine) has been very effective for many patients.6,7 Many oral medications used for neuropathic pain are also commonly used in this disorder. These include tricyclic antidepressants, serotonin and norepinephrine reuptake inhibitors, anticonvulsants, antiarrhythmic agents, baclofen, and opiates when necessary. Individual patients may respond well to aspirin (particularly those with underlying myeloproliferative disease) and vasoactive agents.8
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








