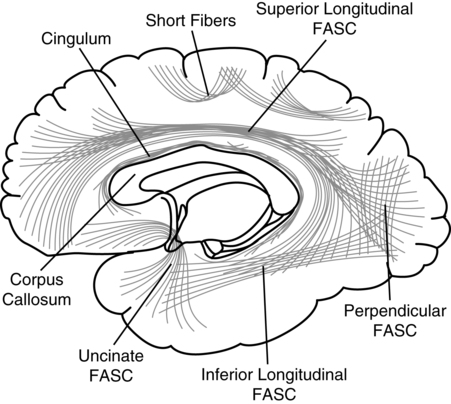
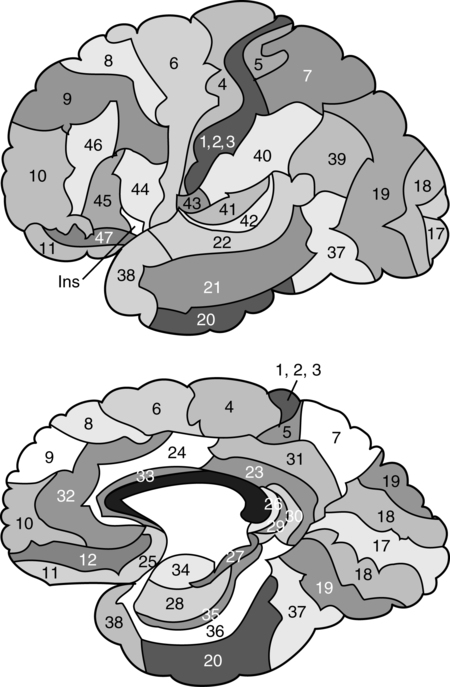
CHAPTER 7 During the late nineteenth and early twentieth centuries, clinical investigators documented diverse behavioral disorders in cases of prefrontal lobe pathology. It was observed that prefrontal pathology did not result in any evident sensory or motor disturbance, but behavioral/personality changes were frequently found. Phineas Gage has become the most typical illustration of frontal lobe dysfunction and has significantly contributed to the understanding of the role of the frontal lobes in behavior. Harlow (1868) described Phineas Gage as a responsible foreman for a railroad company who suffered a tragic accident in which a tampering rod was projected through his frontal lobes. After this accident, profound personality changes were evident, and he was described as “no longer Gage” by associates who perceived his behavior as “profane,” “irascible,” and “irresponsible.” It was of interest to Harlow that cognitive functions (i.e., memory, language, etc.) remained intact, whereas personality (manner of behaving) was so greatly altered. Phineas Gage has become one of the best known classical cases in the history of the neurosciences, and different papers have been devoted to its analysis (e.g., Damasio, Grabowski, Frank, et al., 1994; Macmillan, 2000, 2008). In 1880 Herman Oppenheim coined the term witzelsucht, which was demonstrated by childishness and joking with “alleged” cheerfulness (Oppenheim, 1890, 1891). The term moria (reflecting “stupidity” and a jocular attitude) was part of the change they observed associated with damage in the prefrontal regions of the brain. Oppenheim’s patients all had tumors involving right frontal areas, frequently invading the mesial and basal areas. Jastrowitz (1888) further noted unconcern and “inappropriate cheerfulness” associated with right frontal pathology. The term “executive functions” is a relatively new term in the neurosciences, and until recently, the preferred term was “frontal lobe functions” (or “prefrontal functions”). “Frontal lobe syndrome” was conceptualized by Feuchtwanger in 1923. He correlated frontal lobe pathology to behaviors that were not related to overt speech, memory, or sensorimotor deficits. He emphasized the personality changes in motivation, affective dysregulation, and the incapacity to regulate and integrate other behaviors. During the following years, particularly during the 1980s and 1990s, a diversity of books specifically devoted to the analysis of frontal lobe syndrome were published (e.g., Fuster, 1989; Levin, Eisenberg & Benton, 1991; Miller & Cummings, 1998; Perecman, 1987; Pribram & Luria, 1973; Stuss & Benson, 1986). Luria (1980) can be regarded as the direct antecessor of the term “executive functions.” He distinguished three functional units in the brain: (1) arousal-motivation (limbic and reticular systems); (2) receiving, processing, and storing information (postrolandic cortical areas); and (3) programming, controlling, and verifying activity (frontal lobes). Luria mentions that this third unit has an executive role. Lezak (1983) used the term “executive functions” to discriminate cognitive functions from the “how” or “whether” of human behaviors. Lezak emphasized the fluid nature of executive functioning and how dependent the cognitive and emotional aspects of functioning were on the “executive.” Baddeley (1986) grouped these behaviors into cognitive domains that included problems in planning, organizing behaviors, disinhibition, perseveration, reduced fluency, and initiation. Baddeley also coined the term “dysexecutive syndrome.” The definition of executive function is encompassed by actions fueled by conceptualizations, such as the ability to filter interference; control attention; engage in goal-directed behaviors; abstracting; problem-solving; metacognition; anticipate the consequences of one’s actions; program motor behavior; inhibit immediate responses; regulate behavior verbally; reorient behavior according to behavioral consequences; perform temporal integration of behavior, personality integrity, and consciousness; and the adaptive concept of mental flexibility (Denckla, 1996; Fuster, 2001; Goldberg, 2001; Grafman, 2006; Luria, 1969, 1980; Miller & Cummings, 1998; Stuss & Benson, 1986; Stuss & Knight, 2002). The concept of morality, ethical behaviors, self-awareness, and the idea of the frontal lobes as manager and programmer of the human psyche are also included. Elliott (2003) defines executive functioning as complex processing requiring the coordination of several subprocesses to achieve a particular goal. Intact frontal processes, although not synonymous with executive functioning, are integral to its function. Although executive functions depend on extended dynamic networks including different brain areas (Koziol & Budding, 2009), it is assumed that the prefrontal cortex plays a major controlling and monitoring role. Neuroimaging results have also implicated posterior, cortical, and subcortical regions in executive functioning (Roberts, Robbins, & Weiskrantz, 2002). Most importantly, the prefrontal cortex does not only participate in those classically recognized executive operations (sequencing, alternating, inhibiting, etc.), but it also plays a core role in coordinating cognition and emotion (Mitchell & Phillips, 2007). Interestingly, most of the disturbances reported in Phineas Gage (and in many cases of prefrontal syndromes) refer to behavioral/emotional disturbances; or more precisely, disturbances in coordinating cognition and emotion/motivation. As noted by Harlow (1868) cognitive functions in Phineas Gage remained intact. The prefrontal lobe has extensive connections to subcortical and limbic system areas (Barbas, 2006; Damasio & Anderson, 2003), and even its orbital portion could be regarded as an extension of the limbic system. Stuss and Alexander (2000) suggest that the most important role of the frontal lobes includes affective responsiveness, social behavior, and personality development. The frontal lobes, particularly the right lobe, have also been related with empathy in general and with “theory of mind”—the ability to attribute mental states to others—in particular (Platek, Keenan, Gallup, & Mohamed, 2004; Stuss, Gallup, & Alexander, 2001). Currently, frontal lobe function research is utilizing functional brain imaging techniques to pool collateral findings, look at antecedents, and use a large sample size to eliminate spurious variables; thus, brain regions that contribute to dysexecutive syndromes may prove to be more multifunctional (Lloyd, 2000). Functional imaging has demonstrated that adults and children with focal, especially frontal right-hemispheric, lesions display similar behaviors such as attentional deficits, inability to inhibit a response, and impersistence of activity (Filley, Young, Reardon, & Wilkening, 1999). Typically, executive functions are analyzed in experimental conditions using diverse research strategies, such as solving diverse problems, finding similarities between two words, providing an answer that requires inhibiting another, etc. A paradigm is created, and the subject is required to solve it. Brain activity can be recorded simultaneously, using brain electrical activity or recording the regional level of activation (e.g., Osaka, Osaka, Mondo, et al., 2004). Alternatively, executive functions are analyzed in brain-damaged populations in order to find the contribution of different brain systems (e.g., Jacobs, Harvey, & Anderson, 2007). This last approach represents the classical neuropsychological method. Executive functions, however, are rarely analyzed in natural ecological conditions. Anatomically, the frontal lobes are the largest lobes of the brain. Laterally, they are anterior to the Rolandic fissure and superior to the Sylvian fissure. Medially, they extend forward from the Rolandic fissure and the corpus callosum. The frontal lobes include (a) the posterior regions of the frontal cortex (agranular frontal cortex), associated with motor activity. They correspond to the primary motor area (Brodmann’s area—BA—4, or the precentral gyrus), on one hand; and the premotor area (or motor association area: BA6, 8—frontal eye field, and BA44—Broca’s area), on the other. And (b) the prefrontral cortex (or granular frontal cortex), corresponding to BA9, 10, 11, 12, 24, 32, 45, 46, and 47, as illustrated in Figure 7-1. The prefrontal cortex is usually subdivided into the dorsolateral, mesial, and orbital regions. The limbic components of the frontal lobe include the anterior cingulum and the posterior section of the frontal orbital cortex (Damasio & Anderson, 2003; Fuster, 2008; Mesulam, 2002). The frontal lobe increases in size throughout phylogenetic evolution. Given the overall size of the human brain, the entire frontal lobe of humans is approximately as large as expected for a primate brain (Semendeferi, Lu, Schenker, & Damasio, 2002), yet two portions of the frontal lobe (the primary motor and premotor areas) are significantly smaller; consequently, the prefrontal area is larger than expected (Schoenemann, 2006). The human prefrontal cortex is much larger than that in pongids (chimpanzees, gorillas, and orangutans): 12.7% of total brain volume, compared with an average of 10.3% for pongid species. Differences are observed in the white matter rather than in the gray matter. Comparing humans (brain size about 1350 cm2) with chimpanzees (brain size about 311 cm2), prefrontal gray volumes are 4.8 times larger in humans, whereas nonprefrontal gray volumes are only 4.2 times larger. However, prefrontal white volumes are about 5.0 times larger in humans, whereas nonprefrontal white volumes are only 3.3 times larger (Schoenemann, Sheehan, & Glotzer, 2005). The prefrontal areas of the frontal lobes can be regarded as association areas or intrinsic cortical areas. Luria (1980) considers that the prefrontal regions correspond to tertiary areas (which participate in processing information of various types) of the cerebral cortex. The prefrontal lobes maintain extensive connections, particularly with other cortical areas, the limbic system, the cortical and subcortical motor areas, and the sensory cortex. The major cortical connections are established with the visual, auditory, and somatosensory cortexes. The prefrontal cortex is also connected with the premotor cortex, and through this, with the primary motor cortex. Some projections are unidirectional (e.g., the caudate nucleus and the putamen); and others appear to be bidirectional (e.g., the dorsomedial nucleus of the thalamus) (Damasio & Anderson, 2003). There are multiple intracortical connections, including the superior longitudinal fasciculus—the main bundle of fibers between the posterior and anterior regions of the cerebral cortex. The uncinate fasciculus connects the anterior temporal lobe with the frontal lobe. The orbitofrontal limbic and mesial frontal cortexes receive projections from the superior temporal gyrus, and the orbitofrontal region receives projections from the inferior temporal cortex. The cingulum connects the frontal lobe with the parahippocampal gyrus. The arcuate fasciculus borders the insula and connects the inferior frontal and medial gyri with the temporal lobe. The occipitofrontal fasciculus extends posteriorly from the frontal lobe to the temporal and occipital lobes (Figure 7-2). According to Damasio and Anderson (2003), it is possible to distinguish the following types of fronto-subcortical connections: Some disagreement exists around the question of whether or not there is a single unitary factor accounting for the diversity of executive functions (e.g., Grafman, 2006; Kimberg, D’Esposito, & Farah, 1997; Stuss & Alexander, 2007). Friedman and colleagues (2008) found that executive functions are highly correlated, suggesting a common factor that goes beyond general intelligence. These authors concluded that executive functions represent one of the most heritable psychological traits. It is not evident, however, what the particular unitary factor saturating the different executive function tests could be. Some different proposals and interpretations have been presented during recent years. Behavior inhibition has been considered as a potential candidate for the single factor responsible for successful performance in different executive tests (Barkley, 1997) alone or in combination with working memory (Pennington & Ozonoff, 1996). Salthouse (1996, 2005), on the other hand, suggested that reasoning and perceptual speed represent the underlying factors related to all executive functions. Salthouse (2005) observed that performance on two common tests of executive functioning, the Wisconsin Card Sorting Test and the Controlled Oral Word Association Test, were strongly correlated with reasoning ability and perceptual speed. Other researchers challenge the existence of such a unitary factor. Thus, some authors have emphasized that certain frontal lobe patients perform well on some tests purported to assess executive abilities but not on others (Godefroy, Cabaret, Petit-Chenal, et al., 1999). Furthermore, it has been reported that correlations among different executive tests are frequently moderate or low and very often lack statistical significance (Salthouse, Atkinson, & Berish, 2003). Some other investigators have taken an intermediate position. For instance, Miyake, Friedman, Emerson, et al. (2000) studied three often-postulated aspects of executive functions (shifting, updating, and inhibition) and concluded that, although these functions are clearly distinguishable, they do share some underlying commonality. Based on the results of their study, the authors stated that executive functions are ”separable but moderately correlated constructs” thus suggesting both unitary and nonunitary components of the executive system. By the same token, several authors have suggested different subcomponents of executive functions (e.g., Anderson, 2001; Delis, Kaplan, & Kramer, 2001; Denckla, 1994; Elliott, 2003; Hobson & Leeds, 2001; Lafleche & Albert, 1995; Piguet, Grayson, Browe, et al., 2002). Thus, Stuss and Alexander (2007) refer to three separate frontal attentional processes within the executive category: energization (superior medial), task setting (left lateral), and monitoring (right lateral). Clinical and experimental research has converged to indicate the fractionation of frontal subprocesses and the initial mapping of these subprocesses to discrete frontal regions (Stuss & Levine, 2002). Factor analysis has also supported that executive functions include several subcomponents (Mantyla, Carelli, & Forman, 2007; Stout, Ready, Grace, et al., 2003). Most frequently, three different prefrontal syndromes associated with specific disturbances in executive functions are separated (Box 7-1). Cummings (1993) indicated that the dorsolateral circuit is the most important to executive functioning. The most noted deficit is an inability to organize a behavioral response to novel or complex stimuli. Symptoms are on a continuum and reflect the capacity to shift cognitive sets, engage existing strategies, and organize information to meet changing environmental demands. Dysfunction in this region disrupts essential component cognitive processes, including working memory and inhibitory control (Anderson & Tranel, 2002). Various researchers, including Luria (1969), have noted preservation, stimulus-bound behavior, echopraxia, and echolalia. According to Fuster (1997, 2002), the most general executive function of the lateral prefrontal cortex is temporal organization of goal-directed actions in the domains of behavior, cognition, and language. Lateral differences are observed: whereas left prefrontal damage is more directly associated with cognitive processes, right damage is associated with both restriction of affect and emotional dyscontrol and defects in the perception or comprehension of emotional information. Anosognosia, impaired empathy, and defects in the appreciation of humor (Shammi & Stuss, 1999) are also found. Following lesion to the right dorsolateral area, a transcortical motor aprosodia is expected, whereas a left-sided dorsal lesion will produce a decline in verbal fluency on word-generation tasks and so-called extrasylvian (transcortical) motor aphasia. A hierarchical model of prefrontal function has been proposed in which dorsolateral and frontopolar regions are serially recruited in a reasoning or memory task that requires evaluation of internally generated information: whereas the dorsolateral prefrontal cortex is involved when externally generated information is being evaluated, the frontopolar area becomes recruited when internally generated information needs to be evaluated (Christoff & Gabrieli, 2000). The anterior cingulate is the origin of the anterior cingulate-subcortical circuit. Goldman-Rakic and Porrino (1985) identified input from BA24 to the ventral striatum, which includes the ventromedial caudate, ventral putamen, nucleus accumbens, and olfactory tubercle. Damage to these circuits causes apathy or abulia (a severe form of apathy). Acute bilateral lesions in the medial frontal area can cause akinetic mutism, in which the individual is awake and has self awareness but does not initiate behaviors. These patients demonstrate diminished drive. The spectrum can range to the extreme following bilateral lesions (i.e., patients can be profoundly apathetic, may rarely move, may be incontinent, may eat only when fed, and may speak only in monosyllables when questioned). They are not emotionally reactive, even with painful stimuli, and appear completely indifferent (Damasio & Damasio, 1989). Subcortical deficits, as seen with Parkinson disease and Huntington disease as well as thalamic lesions, may cause apathy if the anterior cingulate is affected. Orbitofrontal syndrome has been associated with disinhibition, inappropriate behaviors, irritability, mood lability, tactlessness, distractibility, and loss of import to events. Affect may become extreme with moria (an excited affect) or Witzelsucht (the verbal reiteration of caustic or facetious remarks), first noted by Oppenheim (1890, 1891). Individuals with this syndrome are unable to respond to social cues, and they are stimulus bound. Cummings (1993) noted that automatic imitation of the gestures of others may occur with large lesions. Interestingly, it has been noted that these patients have no difficulty with card-sorting tasks (Laiacona et al., 1989). Eslinger and Damasio (1985) coined the term “acquired sociopathy” to describe dysregulation that couples both a lack of insight and remorse regarding these behaviors. Much of this may reflect the stimulus-bound nature of this disorder. The orbitofrontal cortex appears to be linked predominantly with limbic and basal forebrain sites. The orbital prefrontal cortex may have the ability to maintain its own level of functional arousal due to its cholinergic innervation from the basal forebrain (Mesulam, 1986). According to Fuster (2002), the ventromedial areas of the prefrontal cortex are involved in expression and control of emotional and instinctual behaviors. The three major prefrontal syndromes could be grouped into two. Ardila (2008) suggested that the prefrontal lobe participates in two closely related but different executive function abilities: (1) “metacognitive executive functions”: problem solving, planning, concept formation, strategy development and implementation, controlling attention, working memory, and the like, which are related with the activity of the dorsolateral prefrontal cortex; and (2) “emotional/motivational executive functions”: coordinating cognition and emotion/motivation (that is, fulfilling biological needs according to some existing conditions), which are related with the orbitofrontal and medial frontal cortexes. Ardila (2008) suggested that “metacognitive” and “emotional/motivational” executive functions may have presented different evolutionary patterns during human phylogeny; and while primates and hominids may possess the second, the first one is only observed in recent human evolution. In the prefrontal cortex, as in other cortical areas, lateralization is observed. Language-related disturbances (such as extrasylvian or transcortical motor aphasia) are more frequently found in cases of left frontal pathology, whereas social, spatial, and, in general, non–language-related disturbances are usually associated with right hemisphere damage. Goldberg (2001) describes two types of cognitive control: one guiding behavior by internal cues and the other by external cues. Normally operating in concert, damage to the frontal lobes can result in perseveration (disinhibited repetition) due to the following: diminished ability to switch behaviors in response to changing demands and environmental dependency, and inability to generate behaviors that are guided and personal. The left prefrontal system is thought to subserve the guiding of cognitive selection by working memory and internal contingencies, whereas the right prefrontal area mediates guiding cognitive selection by external environmental contingencies. Even though executive dysfunction that follows focal brain injury most often occurs (or is most severe) following frontal lobe injury, not all executive processes are exclusively sustained by the frontal cortex (Andres & Van der Linden, 2002). Lesions in nearly any part of the brain have been associated with executive dysfunction (Hausen, Lachmann, & Nagler, 1997). Contemporary research even finds strategy operations in the occipital cortical neurons on visual tasks (Super, Spekreijse, & Lamme, 2001). Andres (2003) analyzed two executive processes: inhibition and dual-task management. He concluded that (1) executive processes involve links between different brain areas, not exclusively with the frontal cortex, (2) patients with no evidence of frontal damage may present with executive deficits, and (3) patients with frontal lesions do not always show executive deficits. A diversity of disorders in communication ability can be observed in cases of frontal lobe pathology including dysarthria, aphasia, language pragmatic disturbances, metalinguistic skill abnormalities, and verbal reasoning impairments. Complex and conceptual verbal abilities may be significantly impaired (Novoa & Ardila, 1987). Most frequently these disorders are found in cases of left hemisphere pathology. The idiosyncrasies of the disorders depend on the specific location and extension of the damage. Alexander, Benson, and Stuss (1989) proposed a comprehensive classification of communication disorders observed in frontal lobe pathology (Table 7-1). Table 7-1 Communication Disorders Associated with Frontal Lobe Pathology Adapted from Alexander, M. P., Benson, D. F., & Stuss, D. T. (1989). Frontal lobes and language. Brain and Language, 37, 656–691. Aphemia was the initial name used by Broca to refer to the impairment in language production associated with left posterior frontal damage (Broca, 1861), but this name was later replaced with aphasia by Trousseau in 1864. The term aphasia prevailed and aphemia was forgotten. During the following decades, the term aphemia appeared from time to time in the neurological literature to refer to the articulatory defects associated with Broca’s aphasia. Schiff, Alexander, Naeser, and Galaburda published an influencial paper in 1983 reacquiring the term aphemia to name the dysarthria following the appearance of left frontal-lobe lesion, including the pars opercularis, inferior prerolandic gyrus (cortical dysarthria), or the white matter deep to those regions. Today, this is the most frequent use of the term aphemia: Aphemia is the spastic dysarthria observed in cases of damage of the upper motor neuron in the pyramidal system. This dysarthria is usually associated with Broca’s aphasia, and it is also observed in cases of damage involving the internal capsule. It is usually recognized that Broca’s aphasia has two different distinguishing characteristics: (a) a motor-articulatory component (lack of fluency, disintegration of speech kinetic melodies, verbal-articulatory impairments, etc.) that is usually referred to as apraxia of speech; and (b) agrammatism (e.g., Benson & Ardila, 1996; Berndt & Caramazza, 1980; Goodglass, 1993; Kertesz, 1985; Luria, 1976). Indeed, a large part of the frontoparietotemporal cortex has been observed to be involved with syntactic-morphological functions (Bhatnagar, Mandybur, Buckingham, & Andy, 2000). Apraxia of speech has been specifically associated with damage in the left precentral gyrus of the insula (Dronkers, 1996; but see Hillis, Work, Barker, et al., 2004).
The executive functions in language and communication
Defining executive functions
The anatomy of the frontal lobes
Intracortical connections
Subcortical connections
Projections to the claustrum, subthalamic region, and mesencephalon
Major dysexecutive syndromes: normal and abnormal conditions
Dorsolateral syndrome
Medial frontal lobe
Orbitofrontal syndrome
Communication disorders in frontal lobe pathology
Left Hemisphere
Right Hemisphere
Lower motor cortex and posterior operculum
Aphemia
Dysprosody
Full operculum plus lower motor cortex
Broca’s area aphasia
Dysprosody
Dorsolateral frontal
Transcortical motor aphasia
Defective pragmatic discourse
Medial frontal
Mutism
Decreased output
Prefrontal
Reduced formulation; impoverished discourse
Disordered formulation; tangential discourse; confabulation
Left hemisphere pathology
Aphemia
Broca’s aphasia
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


The executive functions in language and communication