Treatment of Cervical Myelopathy: Laminoplasty
Eiji Wada
Kazuo Yonenobu
The dorsal approach to the cervical spine was the primary method of access to the spinal canal until the ventral approach was developed by Robinson and Smith (1) and Cloward (2). Dorsal decompression was performed by removing the spinous processes, laminae, and articular processes with a rongeur. With the accumulation of experience in dorsal decompression for cervical spondylotic myelopathy (CSM), successful laminectomy was possible only when lordotic alignment of the cervical spine, wide and extensive laminectomy for adequate dorsal shift of the spinal cord (3), and stability of the spine (4,5) were secured after surgery. The thick scar formation occasionally resulting after laminectomy hematoma could lead to an unfavorable outcome.
The insertion of surgical instruments, such as a Kerrison rongeur or a curette, into the spinal canal without awareness of canal narrowness, or uneven decompression of the spinal cord during resection of the laminae, can impinge or distort the spinal cord and result in deterioration of neurologic function. Several investigators have indicated the hazard of postoperative loss of neural function due to surgical intervention (6, 7 and 8).
Due to the poor results of conventional laminectomy for cervical compression myelopathy, Kirita (9) and Miyazaki and Kirita (10) developed extensive simultaneous decompression laminectomy to avoid distortion of the spinal cord by the edges of the resected laminae.
Oyama et al. (11) devised an expansive Z-shaped laminoplasty, in which the dorsal wall of the spinal canal was preserved by Z-plasty of the thinned laminae. They thus attempted to prevent the invasion of scar tissue, that is, the so-called laminectomy membrane, which was believed to be a cause of late neurologic regression. They also anticipated that the laminae reconstructed by Z-plasty would support for the spine. The introduction of a high-speed air drill allowed the successful development of this procedure.
In 1977, Hirabayashi et al. (12) introduced an epochmaking laminoplasty, the expansive open-door laminoplasty. They described the advantages of this procedure: multiple levels of the spinal cord can be decompressed simultaneously, better postoperative support of the neck allows earlier mobilization of the patients, postoperative kyphotic deformity of the cervical spine can be prevented, and mobility of the cervical spine is reduced postoperatively, which helps to prevent late neurologic deterioration as well as the progression of ossification of the posterior longitudinal ligament (OPLL).
After the introduction of the laminoplasty of Hirabayashi, various modifications and supplementary procedures have been devised for further improvement of safety and efficacy of decompression and for stability of the spine.
AIMS, ADVANTAGES, AND DISADVANTAGES OF LAMINOPLASTY
The aims of the laminoplasty are to expand the spinal canal, to secure spinal stability, and to preserve the protective function of the spine. Preservation of mobility of the spine is also the goal of this procedure for multiple-level involvement.
The maximum expansion of the canal is obtained at its central part in the bilateral hinge type of laminoplasty and at its opening side in the unilateral hinge type. Dorsal migration of the spinal cord after both types of laminoplasty has been confirmed using computed tomography (CT) myelography (13,14). Sodeyama et al. reported that the spinal cord moved a mean of 3 mm (0 to 6.6 mm) dorsally at C5 or C6. In the unilateral hinge type, decompression can be extended along the nerve root on the opening side by partial facetectomy.
Preservation of the dorsal spinal structures permits reinsertion of the nuchal muscles and the spinal ligaments after they have been completely or partially detached. This prevents kyphosis or listhesis of the cervical spine, which often develops after laminectomy, particularly in subjects aged less than 50 years. Reattaching muscles or ligaments to the spinous processes after the laminoplasty may improve the dynamic stability of the spine.
The development of kyphosis and a thick peridural scar, which develops subsequent to laminectomy, is a notorious cause of late neurologic worsening. The spared laminae
preserve the protective function of the spine, shielding the spinal cord from pressure of hematoma during the early postoperative periods and preventing the invasion of scar tissue in the late convalescent period.
preserve the protective function of the spine, shielding the spinal cord from pressure of hematoma during the early postoperative periods and preventing the invasion of scar tissue in the late convalescent period.
Advantages of laminoplasty include the fact that the spinal cord can be decompressed without removal of spondylotic protrusion impinging on the neural tissue. Removal of the osteocartilaginous protrusion encroaching on the already compromised neural tissue is the most hazardous portion of the procedure when surgeons use the ventral approach to CSM. A second advantage is that laminoplasty facilitates obtaining hemostasis around the affected spinal cord. Ventral surgery for CSM and OPLL often demands more careful and meticulous procedures for hemostasis. These procedures may threaten the spinal cord or the nerve root that is already distressed. A third advantage is that laminoplasty allows additional procedures for nerve root decompression or reinforcement of spinal stability. Partial facetectomy for nerve root decompression is optional except for the facets on the hinge side of the laminae. Bone grafting for stabilization, in either single or multiple segments, is readily applicable.
There are two disadvantages of laminoplasty. First, range of motion (ROM) of the cervical spine is reduced, particularly in extension, lateral bending, and rotation. This becomes more marked when laminotomy or hinges were located at the facet in either expansive open-door laminoplasty or spinous process-splitting laminoplasty. Second, discomfort of the neck may result after laminoplasty. This is one of the most discouraging complications. Not placing a gutter for the hinge directly at the facet, avoiding too much muscle detachment during surgery, and encouraging mobilization of the neck as early as possible are all believed to lessen the postoperative discomfort (15).
INDICATIONS AND CONTRAINDICATIONS FOR LAMINOPLASTY
Generally, laminoplasty can be indicated for myelopathy secondary to developmental spinal canal stenosis (AP canal diameter <13 mm), continuous or mixed type of OPLL, and multisegmental spondylosis associated with a narrow spinal canal.
Laminoplasty should be considered for patients who have multisegmental spondylotic myelopathy with a relatively narrow spinal canal (an AP canal diameter of 13 to 14 mm) (16). When this condition is associated with spinal instability, laminoplasty should be supplemented by spinal arthrosis.
For one- or two-level spondylosis without developmental spinal canal stenosis, anterior discectomy and arthrodesis is the treatment of choice.
In cervical disk herniation, the consensus has been that anterior discectomy and spinal fusion (ASF) is preferred for one or two vertebral segments. However, for patients with narrowed spinal canals (<13 mm), laminoplasty has had favorable results because it avoids adjacent segment degeneration and bone graft complications. Resorption of herniated disk fragments has been confirmed in follow-up MRI studies (17).
Spinal cord tumors can also be extirpated by laminoplasty in combination with ultrasonic suction. For younger patients, laminoplasty should be borne in mind. Laminoplasty is preferable to laminectomy because of the lower risk of postoperative kyphosis and instability. Laminoplasty, with stabilization (arthrodesis), has been suggested for myelopathy secondary to multilevel subaxial subluxation in patients with rheumatoid arthritis.
At present, no type of laminoplasty can correct a fixed kyphotic deformity to a lordotic curve. Accordingly, a kyphotic cervical spine is a contraindication for laminoplasty. However, laminoplasty, combined with a formal arthrodesis procedure, can be indicated for myelopathy or radiculopathy secondary to multisegmental spondylosis associated with athetoid cerebral palsy, provided that the athetoid movements of the neck can be adequately controlled with a halo vest during the postoperative period.
The age of the patient does not place a limit on laminoplasty because the invasiveness of the procedure is similar to that of laminectomy. Laminoplasty is beneficial, even in patients more than 75 years of age, in improving neurologic function and ability to engage in activities of daily living (18). The average operating time in the authors’ series was 2.1 hours, and the average of blood loss was 225 mg. Hirabayashi reported an average operating time of 2 hours and an average blood loss of 362 mL, respectively.
TECHNIQUES OF LAMINOPLASTY AND THE SUPPLEMENTARY PROCEDURES
The basic concept of each surgical procedure is similar to that of the expansive open-door laminoplasty already described. Therefore, this procedure is described in more detail. Further details of other procedures should be obtained from the original articles (19, 20 and 21). No procedure has been proven to be statistically rostral to any others with regard to the neurologic and roentgenographical results.
The patient is placed in the prone position on a laminectomy frame to decrease the abdominal pressure. A threepoint pin fixation device such, as Mayfield fixation, is strongly recommended to secure the head and maintain cervical alignment in the neutral position or in slight extension. When spinal arthrodesis is required, the cervical spine is adjusted to its proper alignment after laminoplasty and prior to fusion.
Through a dorsal midline incision, the nuchal ligament is divided at the midline. Typically with CSM, the extent of decompression is from C3 to C7 for complete dorsal migration of the spinal cord. Decompression should also be wide enough for the spinal cord to migrate dorsally. However, surgeons differ regarding placement of the laminotomy for the opening and the gutter for the hinge. Both of these factors are believed to be related closely to the nature and incidence of the complications of this procedure.
UNILATERAL HINGE LAMINOPLASTY
Expansive Open-Door Laminoplasty of Hirabayashi
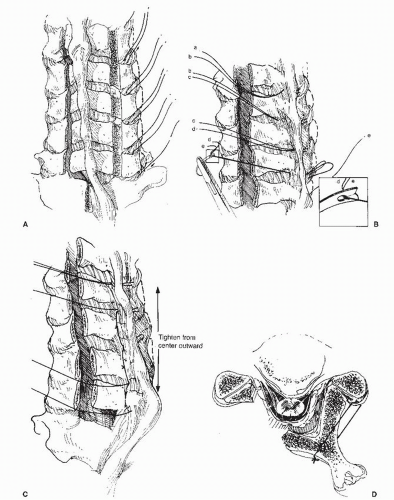
The spinous processes are exposed from C2 to T1, with care taken not to damage the supraspinous and interspinous ligaments (19) (Fig. 77.1). A gutter is then created at the junction of the articular processes and the
laminae with a steel burr followed by a diamond burr. The laminae are then cut along the gutter with a thinbladed Kerrison rongeur. Next, another gutter is created on the hinge side with a steel burr, being set more laterally than the gutter on the opening side. Opening of the laminae is secured by sutures placed on the facet joint capsules on the hinge side and the corresponding laminae.
laminae with a steel burr followed by a diamond burr. The laminae are then cut along the gutter with a thinbladed Kerrison rongeur. Next, another gutter is created on the hinge side with a steel burr, being set more laterally than the gutter on the opening side. Opening of the laminae is secured by sutures placed on the facet joint capsules on the hinge side and the corresponding laminae.
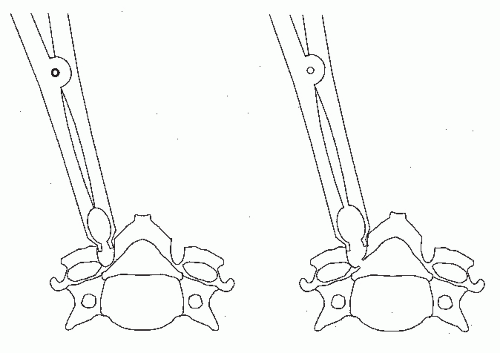
If the lifted laminae are not fixed firmly, the enlargement of the spinal canal may be reduced (lamina closure). A bone graft used to support the lifted lamina was devised by Itoh and Tsuji (20) to avoid loss of enlargement in the so-called en-bloc laminoplasty. Use of a Kerrison rongeur can present another pitfall because insertion of even a thin-bladed rongeur into the spinal canal may cause spinal cord or vascular (epidural venous plexus) injury and lead to neurologic deficits or heavy bleeding. We use a steel burr to cut the outer cortex and cancellous bone of the laminae and then thin the inner cortex progressively with a diamond burr. With adequate irrigation and suction, the color of the cortex can be seen to change from ivory to dark red as the epidural venous plexus becomes evident. Because the cranial part of each lamina is thicker than the caudal part and is covered by the caudal portion of the lamina above, it requires more grinding than the caudal part to equalize the thickness of the inner cortex. Then, a scalp clip holder is inserted into the gutter and opened to separate both edges of the gutter by fracturing the thinned inner cortex (Fig. 77.2). With this technique, no instrument is inserted into the spinal canal.
En-bloc Laminoplasty of Itoh and Tsuji
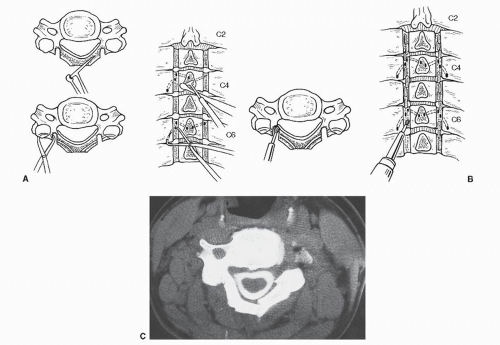
Through a conventional dorsal midline approach, the spinous processes are cut at their bases and kept for bone grafting (20). Two gutters are made in the laminae just medial to each articular process. Tunnels for wiring are made within the laminae and the caudal articular processes at C4 and C6 using a specially designed awl and pusher. After the en-bloc lifting of the laminae, usually from C3 to C7, grafts made of the resected spinous processes are placed between the articular processes and the edges of laminae at C4 and C6 to support the lifted laminae. The laminae and bone grafts are then secured in position with steel wires (Fig. 77.3).
Foraminotomy on the opening side can be combined with this technique. However, wiring for fixation of the laminae and grafts complicates this procedure and also makes it difficult to obtain postoperative MRI scans. The authors use a nonabsorbable synthetic suture material for fixation and place sutures only on the opening side unless the hinges are stable.
Other modifications to prevent lamina closure:
Suture Anchor: Wang described the use of suture anchor (GII anchor, Mitek, Westwood, MA) to avoid displacement of elevated lamina or the reclosure of the opening (22). As, in his series of 12 patients, the anchoring systems did not fail during the follow-up period (mean 29.5 months) and the decompression was maintained, he concluded that the use of anchor systems to stabilize the dorsal elements after laminoplasty is a simple and effective technique for maintaining the increased sagittal diameter of the canal. Following his report, several instruments (AO cortical screw with nonabsorbable sutures, titanium-alloyed screw with nonabsorbable sutures or Mitek Tacit QuickAnchor suture anchors) have been reported to be cost-effective. On the other hand, Matsumoto reported that the use of anchor screws was not significantly correlated with the occurrence of lamina closure (23).
Mini Plate: Frank and Keenen (24) reported the laminoplasty technique using titanium miniplates that successfully reconstructs an enlarged rigid spinal canal. Recently, a specially designed miniplate system to fit the contour of the laminae was introduced. Although plate and screw systems have the advantages of a greater rigid fixation than traditional technique, they are arguably technically difficult to place because the laminae or the facets are generally small in size.
BILATERAL HINGE LAMINOPLASTY
Spinal fusion can also be performed with this bilateral hinge laminoplasty, but foraminotomy is often very difficult to perform.
Spinous Process-Splitting Laminoplasty of Kurokawa
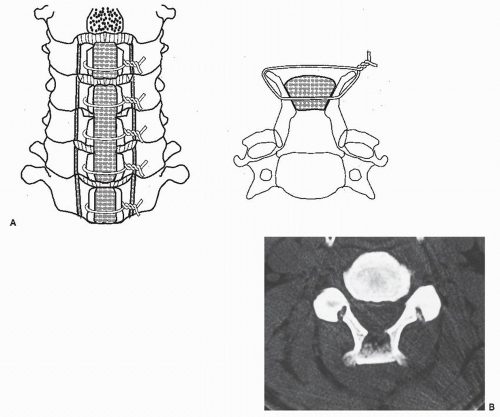
A dorsal approach is made along the nuchal ligament to the line of the spinous processes (Fig. 77.4). The semispinalis cervicis is transiently detached from C2 spinous process. To split C2, the rectus capitis posterior major and obliquus capitis caudal muscles are also detached. Cervical laminae are exposed laterally to the medial aspect of the facet joints, and the interspinous ligaments are removed. The spinous processes are split sagittally with a high-speed burr. After bilateral gutters for the hinges are carefully crafted with a high-speed burr at the transitional area between the facet joint and laminae, spinal canal enlargement is achieved by opening the split laminae bilaterally with a spreader and placing corticocancellous iliac crest autografts held in place with sutures or wire (25). When dorsal spinal fusion is required, one long iliac bone is placed between the spinous processes to be fused, which then are secured with wire. A ceramic spacer can also be substituted for an autogenous bone graft (26).
Tomita modified the process of spinous process splitting by using a thread-wire saw (T-saw laminoplasty) (27). The author reported that this technique shortens the mean time of surgery by 63 minutes and lessens the mean blood loss by 70 mL compared with the original procedure.
PROCEDURES SUPPLEMENTARY TO LAMINOPLASTY
The nuchal muscles and ligaments are believed to be an indispensable structure helping to stabilize the cervical spine in lordosis. In fact, the nuchal muscles are displaced laterally and ventrally in patients who develop kyphotic deformity after laminectomy, indicating that these muscles and ligaments play an important role in stabilization
of the cervical spine. Recent morphologic study by coronal view of MRI also revealed that repair of semispinalis cervicis is important for maintenance of cervical lordosis after laminoplasty (28). Therefore, the following procedures have been performed in an attempt to reconstruct soft tissue for restoration of stability in lordosis.
of the cervical spine. Recent morphologic study by coronal view of MRI also revealed that repair of semispinalis cervicis is important for maintenance of cervical lordosis after laminoplasty (28). Therefore, the following procedures have been performed in an attempt to reconstruct soft tissue for restoration of stability in lordosis.
Reattachment of the Nuchal Muscles to the Spinous Process of the Axis
The rectus major, inferior oblique, and semispinalis cervicis muscles originally attached to the spinous process of the axis are detached from their origins along with small fragments of the spinous process tip, and the lamina and articular processes of C3 are exposed by retracting the semispinalis cervicis muscles laterally (Fig. 77.5). After laminoplasty, the muscles are reattached to their origins by suturing the bony fragments to the tip of the spinous process. The other nuchal muscles are also repositioned and sutured to each other in order to form a suspensory nuchal ligament.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree