It may also result from a lesion in the oculomotor or vestibular systems from diplopia or oscillopsia. Patients with alexia or agnosia may also complain of visual loss, although their acuity may test normally.
TABLE 9.1 Signs and Symptoms that Suggest Location in the Visual Pathway | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
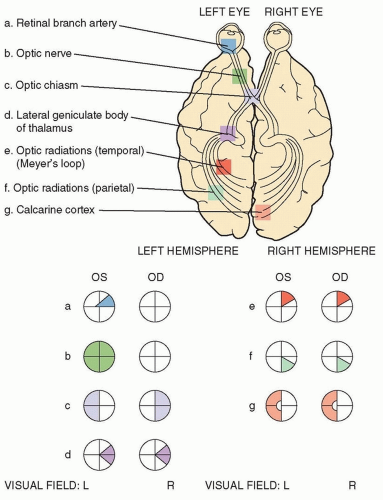
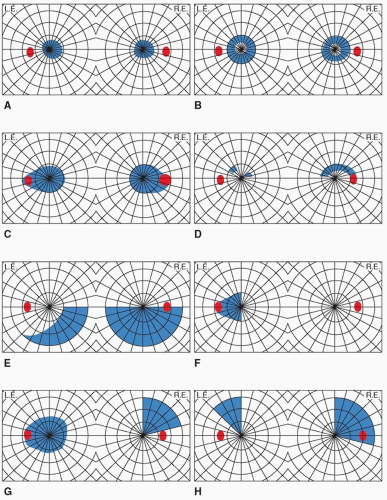
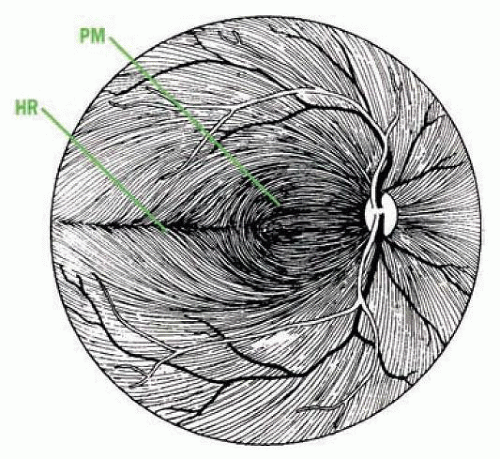
the nasal fovea and axons originating between the fovea and the disc course directly into the temporal disc, and damage to these axons results in a cecocentral scotoma (Fig. 9.2C). This bundle then occupies the center of the optic nerve anterior to the chiasm. Damage to fibers coming from the retina nasal to the disc leads to temporal sectoral defect with an apex at the blind spot.
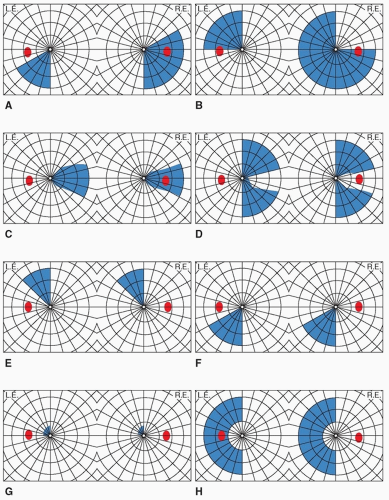
anterior in the occipital lobe, where peripheral vision is represented, results in a monocular temporal crescent visual field defect. After a retinal detachment has been ruled out, this is specific for an occipital lobe lesion contralateral to the field defect and is usually caused by a tumor. Sparing of the anterior calcarine cortex is common in infarcts of the posterior cerebral artery and results in a preservation of the temporal crescent contralateral to the lesion. It is highly suggestive of occipital localization. A homonymous hemianopia with sparing of the central visual field is referred to as macular-sparing hemianopsia (Fig. 9.4H) and is suggestive of occipital involvement but macular sparing has been reported as anterior as the optic tract. Preservation of motion vision in a hemianopic defect, termed the Riddoch phenomenon, is also suggestive of occipital involvement, although it has also been seen as far anteriorly as the optic tract as well. Anton syndrome, denial of blindness in the setting of bilateral hemianopsias, represents bilateral calcarine
involvement plus an association area infarction. Quadrantic visual field defects precisely respecting the horizontal meridian in each eye have been reported in extrastriatal occipital cortex lesions involving areas V2/V3.
TABLE 9.2 Causes of Transient Unilateral Visual Loss | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree