and Kelly Del Tredici1
(1)
Zentrum f. Biomed. Forschung AG Klinische Neuroanatomie/Abteilung Neurologie, Universität Ulm, Ulm, Germany
The pattern of lesions that slowly develops during the AD-process is more easily understood and interpreted against the anatomical backdrop of involved regions and their major connections, which can be depicted in simplified diagrams (Figs. 3.1a, 6.9, and 6.10).
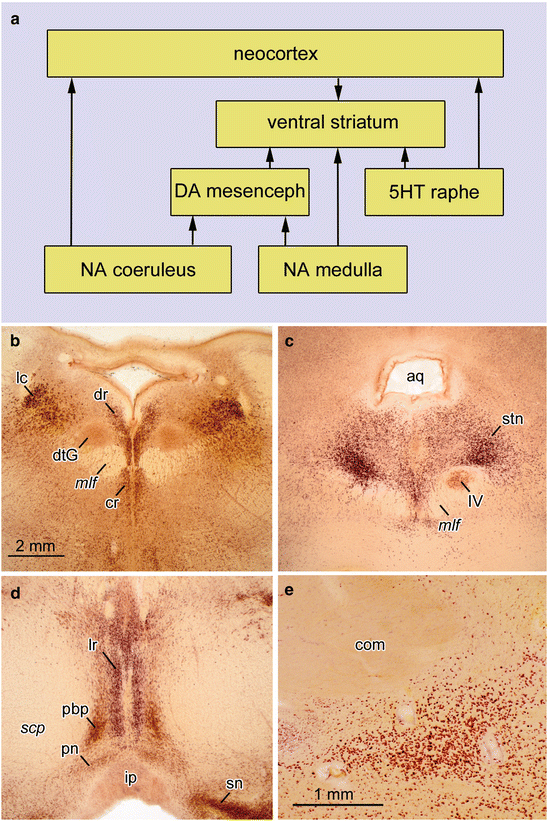

Fig. 3.1
(a) Diagram of interconnections between important non-thalamic nuclei with diffuse projections to the cerebral cortex. (b–e) 400 μm sections cut perpendicularly to the brainstem axis. Pigment-Nissl stain (aldehydefuchsin and Darrow red) from a 53-year-old male control. (b) Locus coeruleus and dorsal raphe nucleus. (c) Supratrochlear portion of dorsal raphe nucleus and motor nucleus of IVth cranial nerve in the medial longitudinal fascicle. (d) Paranigral nucleus, parabrachial pigmented nucleus, and linear raphe nucleus. (e) Basal nucleus of Meynert. Abbreviations: aq—mesencephalic aqueduct; com—anterior commissure; cr—central raphe nucleus; DA mesenceph—dopaminergic neurons, mesencephalon; dtG—dorsal tegmental nucleus of Gudden; dr—dorsal raphe nucleus; ip—interpeduncular nucleus; lr—linear raphe nucleus; mlf—medial longitudinal fascicle; NA coeruleus—noradrenergic neurons, locus coeruleus; NA medulla—noradrenergic neurons, medulla oblongata; pbp—parabrachial pigmented nucleus; pn—paranigral nucleus; scp—decussation of superior cerebellar peduncle; sn—substantia nigra; stn—supratrochlear portion of the dorsal raphe nucleus; 5HT—serotonergic neurons; IV—motor nucleus of the trochlear nerve. Scale bar in (b) is valid for (c) and (d)
A few nuclei within the brainstem, midbrain, basal forebrain, and hypothalamus send long and profusely ramifying projections to the olfactory bulb, the cerebral cortex, many subcortical nuclei (with the notable exception of the pallidum), the cerebellum, and spinal cord. Because of their extensive projections, these nuclei belong to a functionally unified group, the non-thalamic nuclei with diffuse cortical projections, in contrast to the nuclei of the thalamus that project to specific cortical areas (Fig. 6.10).
Alone the fact that the axons of projection neurons from the non-thalamic nuclei have such an unusually large number of ramifications (Fig. 5.3a) indicates that they are not destined to relay data selectively from one site to another, but that their effect on cortical regions is more universal and generalized (Morrison et al. 1982). To this end, such axons share a common hallmark, namely, their terminal branches develop numerous local varicosities equipped only with presynaptic sites (non-junctional varicosities) supplemented by varying amounts of classical synaptic connections that have both presynaptic and postsynaptic sites (Agnati et al. 1995; Nieuwenhuys 1999; O’Donnell et al. 2012). By means of the non-junctional varicosities, the axons release neurotransmitters and neuromodulators diffusely into the interstitial fluid (ISF) (volume transmission), thereby activating receptors of astrocytes, oligodendrocytes, and microglial cells, as well as of cells of the vasculature and nerve cells within a given local diffusion zone (Kalaria et al. 1989; Agnati et al. 1995; O’Donnell et al. 2012). The mechanism of volume transmission permits influence of neurotransmitters and neuromodulators on many cells within a given local diffusion zone.
Non-thalamic nuclei with diffuse cortical projections include not only the noradrenergic locus coeruleus (Fig. 3.1a, b) and related areas but also the serotonergic nuclei of the upper raphe system (Fig. 3.1a–c), the dopaminergic paranigral and parabrachial pigmented nuclei of the mesencephalic tegmentum (Fig. 3.1a, d), the histaminergic tuberomamillary nucleus of the hypothalamus (Panula et al. 1990; Saper 2004), and the cholinergic magnocellular nuclei of the basal forebrain (Fig. 3.1e) (Mesulam 2004).
All of these nuclei belong to the isodendritic core of the brain (Ramón-Moliner and Nauta 1966) and are located far apart from each other. During the later phases of primate evolution when the cerebral cortex increased considerably in size, the nuclei under consideration developed new subnuclei as extensions of their phylogenetically older portions. An example of this trend to ‘integrated phylogeny’ (Rapoport 1988, 1989) in humans is the supratrochlear portion of the dorsal raphe nucleus, which, alone by virtue of its size, is very prominent (Fig. 3.1c) and thereby differs from corresponding subnuclei in the brains of non-human mammals.
The locus coeruleus in the pontine tegmentum contains nearly 32,000 nerve cells and is the largest accumulation of noradrenergic neurons in the human brain (Aston-Jones and Cohen 2005; Szabadi 2013). Mature projection neurons in this nucleus display deposits of neuromelanin granules. The lipofuscin-like matrix of these granules becomes melanized by taking up by-products of the catecholamine metabolism (Double et al. 2008). Their natively brown color makes it possible to identify clusters of melanized neurons located just below the surface of the fourth ventricle, and they are also clearly visible in cross-sections through the brainstem (Fig. 3.1b). The slender column-like principal portion of the locus coeruleus lies close to the lateral angle of the fourth ventricle (Fig. 3.1b lc), commences from the level of the VIIth cranial nerve, and extends rostrally to approximately the level of the decussation of the IVth cranial nerve. The locus coeruleus receives relatively few afferents, some from nearby nuclei (for instance, from the prepositus nucleus of the hypoglossus), whereas other afferents originate in the contralateral locus coeruleus, and important steering projections come from the central subnucleus of the amygdala. Additional afferents have their origins in distant regions, e.g., the modulating projections from dorsal areas of the prefrontal neocortex to the locus coeruleus (Benarroch 2009; Sara 2009).
The subcoeruleus portion underneath the anterior half of the principal portion is less densely packed with melanized neurons and generates mainly descending projections. The flattened cerebellar portion projects to the cerebellum and merges with the principal portion extending along the superior cerebellar peduncle directly underneath the roof of the fourth ventricle. Closely related to the locus coeruleus are more loosely arranged groups of melanized neurons elsewhere in the lower brainstem: one group is located in lateral portions of the intermediate reticular zone. A second group is a component of the dorsal vagal area, and a third group consists of noradrenergic cells surrounding the facial nucleus and the complex of the superior olive (Counts and Mufson 2012).
The serotonergic upper raphe complex comprises the dorsal, central, and linear nuclei. The dorsal nucleus lies between the medial longitudinal fascicles and the ependymal lining of the fourth ventricle (Fig. 3.1b dr). It almost extends as far caudally as the locus coeruleus. Rostrally, it covers the motor nucleus of the trochlear nerve (Fig. 3.1b IV) and forms the expansive supratrochlear subnucleus (Fig. 3.1c stn). The central nucleus lies ventrally from the medial longitudinal fascicles (Fig. 3.1b cr), whereas the linear nucleus extends far into the decussation of the superior cerebellar peduncle (Fig. 3.1d lr).
With the exception of melanized neurons in the substantia nigra, the dopaminergic neurons of the midbrain also belong to this system of non-thalamic nuclei with diffuse cortical projections. The perirubral subnucleus lies close to the red nucleus, while the paranigral nucleus (Fig. 3.1d pn) forms an arch covering the interpeduncular nucleus (Fig. 3.1d ip) and continues into the leaf-like and sagittally-oriented pigmented parabrachial nucleus (Fig. 3.1d pbp) that accompanies the linear raphe nucleus (Fig. 3.1d lr).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







