and Kelly Del Tredici1
(1)
Zentrum f. Biomed. Forschung AG Klinische Neuroanatomie/Abteilung Neurologie, Universität Ulm, Ulm, Germany
7.1 Stages 1a and 1b: Development of Inclusions in Axons and of Pretangle Material in Transentorhinal Pyramidal Cells
As the pathological process progresses and secondary to the subcortical lesions described above, abnormal tau aggregates appear for the first time in the cerebral cortex generally in the transentorhinal region. AT8-ir lesions are present in neuronal processes, most probably in terminal portions of axons, and we designate such cases as having “cortical stage 1a” tau pathology (Table 2.1; Figs. 7.1 and 7.2). The predominantly radially aligned thread-like cellular processes are easier to see in unconventionally thick sections (Fig. 7.1a, b). A thin network of finely shaped and weakly AT8-ir neuronal processes (axons) together with more intensely immunostained dots develops. Sometimes, however, it is possible to follow several of the processes over longer distances, and they often end with a droplet-like swelling (Figs. 7.1c, d and 7.2a). All stage 1a cases show the presence of subcortical tau lesions resembling those found in stages a–c (see Sects. 5.1 and 5.2).
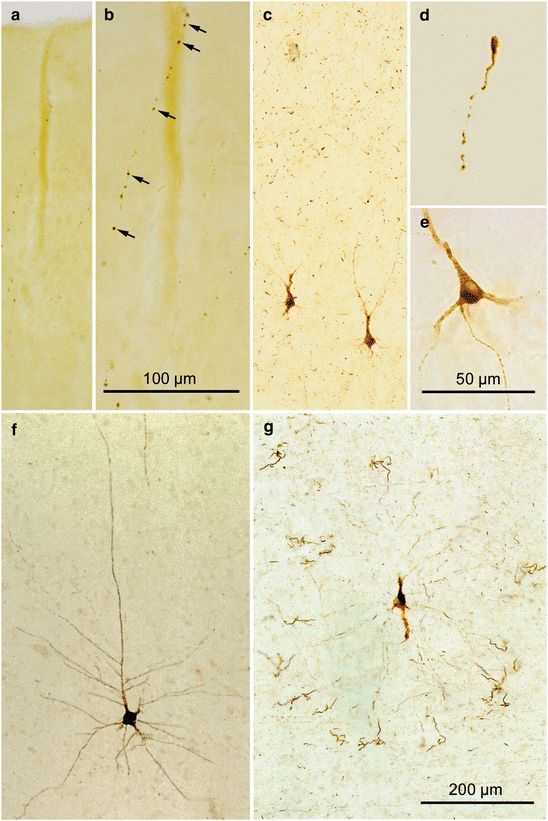
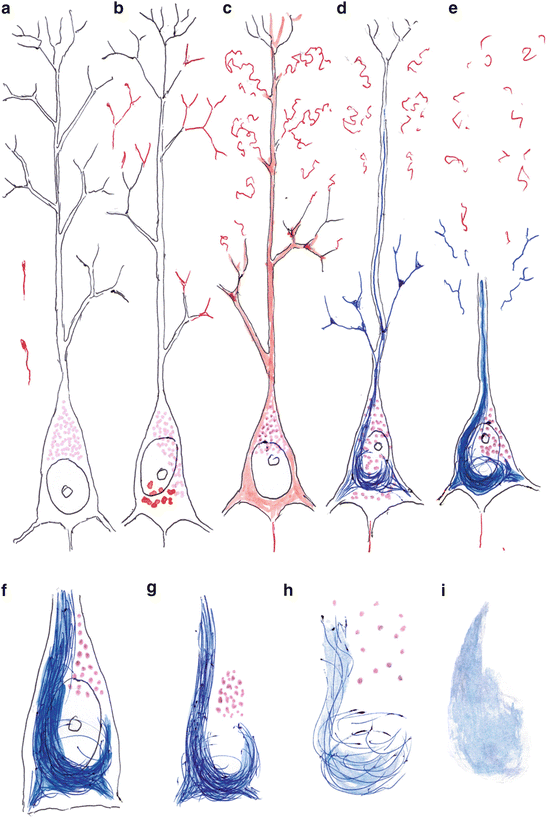

Fig. 7.1
Stages 1a and 1b (cortical AT8-ir but non-argyrophilic tau inclusions) in 100 μm AT8-immunostained sections. (a, b) Stage 1a. (a) At first glance it appears that the transentorhinal cortex has no AT8-ir lesions. Micrograph in (b) shows the region around the cortical vessel seen, in (a), at higher magnification. Arrows point to a single AT8-ir axon, with pathological inclusions appearing like a string of perls. (d) Occasionally, terminal portions of altered axons with knob-like enlargements at their tips can be observed. The immunoreactive material in axons is recognizable in thick sections but easily missed in thin paraffin sections used for routine diagnostics. (c, e–g) Stage 1b. AT8-ir pyramidal cells appear suddenly in addition to the already-existing subtle axonal network (see especially the upper half of c). (e) AT8-ir material is present not only in the somatodendritic domain but also in the axon of the affected pyramidal cells. (f) Initially, the AT8-ir pyramidal cells appear intact and resemble Golgi-impregnations of a healthy nerve cell. (g) Later, however, the distalmost segments of their dendrites become twisted and exhibit varicosities and appendages. Eventually, these tips lose their contact to the proximal branches. Scale bar in (g) also applies to (a, c) and (f)

Fig. 7.2
Schematic drawing of the development of pathological changes in transentorhinal pyramidal cells of layer pre-α. (a) The cortical neuropil displays AT8-ir neuronal processes, some of which show a knob-like enlargement at their tip. The lipofuscin granules (violet stippling) usually lie close to the stem of the apical dendrite. (b) With relative abruptness, AT8-ir material appears intraneuronally in the form of droplets in the soma but also distributed diffusely at branching sites of dendrites. (c) The soluble material fills the entire cell, including the axon. Then, the distal dendritic segments become abnormally altered and detached from the stems. (d) The abnormal tau material in the somatodendritic compartment readily converts into argyrophilic NTs and NFTs (argyrophilic material is depicted in blue), whereas the abnormal material in the axon remains in a non-argyrophilic state. (e) The NFT consolidates and partially extends into dendritic stems. Terminal branches of the dendrites are lost, and the complexity of the dendritic tree is severely reduced. (f) The cell soma of surviving pyramidal cells shows a light nucleus with a large nucleolus. (g) Loss of the cell nucleus is seen easily in thicker sections. The compactness of the NFT gradually decreases, and the lipofuscin granules maintain their position for awhile. (h) After a relatively long period of time, the lipofuscin granules begin to dissipate and they eventually disappear from the tissue. The argyrophilia of the NFT decreases and the NFT loses its compact appearance. (i) The NFT leaves behind a permanent tombstone tangle. Here, AT8-ir material appears in red and Gallyas-positive material is depicted in blue
During cortical stage 1b (Table 2.1, AT8-ir inclusions in cortical pyramidal cells), the stage 1a lesions are accompanied by AT8-ir transentorhinal pyramidal cells that closely resemble the multipolar neurons of the superficial entorhinal layer pre-α. In the transentorhinal region, such multipolar cells slope gradually downwards until they reach the upper surface of layer V (Fig. 6.2). In so doing, they undergo a transformation from star-shaped multipolar cells in layer pre-α into modified pyramidal cells with an apical dendrite that develops gradually (Braak and Braak 1992a).
These modified pyramidal cells develop tau aggregates so suddenly that it is impossible to distinguish any precursors under the light microscope. In contrast to the process in coeruleus neurons, however, abnormal tau material frequently appears initially in dendrites (Fig. 7.2b) and then fills the soma and the axon (Fig. 7.2c) (Braak et al. 1994). It is indeed difficult to comprehend how these modified pyramidal cells could synthesize enough normal tau in the somatodendritic compartment within such a short interval. As such, it is more probable that a production phase precedes the immediately beginning aggregation. Yet, it is unknown what mechanisms trigger the pyramidal cells (e.g., pathological signaling) to commence producing large amounts of normal tau because their somatodendritic compartments do not need the protein. Up to stage 1a, only the brainstem nuclei with diffuse cortical projections display AD-associated intraneuronal lesions. Thus, it is logical to surmise that transentorhinal projections neurons can be reached and influenced by the axonal terminals of these nuclei, although it is admittedly difficult to understand how such a one-to-one contact between a single axon terminal and a single neuron can take place. Since only a small fraction of the newly synthesized tau protein finds binding sites in the somatodendritic compartment, the protein presumably exists in the cytosol, for the most part, in a hyperphosphorylated state. There, after exceeding critical concentrations, it may convert within a short time interval into an irreversibly hyperphosphorylated and slightly aggregated state.
Immediately afterwards, the involved pyramidal cells are nearly filled—reminiscent of a Golgi impregnation—with the pathological material and, initially, they barely deviate from their normal shape (Figs. 7.1f and 7.2c). Next, local swellings appear at the most distal segments of the dendrites, which slowly become curled and twisted (Figs. 7.1g and 7.2c). With time, the terminal dendrites develop short appendages and then look as if they are isolated in the neuropil without any connection to their proximal portions (Figs. 7.1g and 7.2c) (Braak et al. 1994). Notably, the distal dendritic segments of cortical pyramidal cells are phylo- and ontogenetically late-appearing structures that chiefly receive axonal contacts of late-emerging and late-maturing pyramidal cells. The functions of these contacts are not known. In any case, loss of the most distal dendritic segments does not impinge on the survival of the involved neurons. At this early stage, the lost dendritic segments contain AT8-ir material that has not converted into argyrophilic inclusions (NTs). The detached dendritic segments are rapidly degraded and leave behind no remnants. At present, it is still unknown whether the abnormal material becomes a component of the interstitial fluid (ISF) and, subsequently, of the cerebrospinal fluid (CSF).
A similar procedure occurs in all of the different types of nerve cells that are susceptible to the AD process. That is, the somatodendritic compartments of these cells pass through the previously described pretangle phase before developing argyrophilic filaments in their somata (NFTs) and dendrites (NTs). The potential for reversing the pathological process is probably highest during the pretangle phase.
7.2 NFT Stages I and II
After passing through stage 1b of the pretangle phase, involved nerve cells begin, for the first time, to produce argyrophilic lesions at branching points of their dendrites. In a further development, the argyrophilic material extends into other portions of the dendrite and forms NTs (Togo et al. 2004). It is still fully unclear why, as a rule, the development of NTs in pyramidal cells precedes that of NFTs. A variety of flame-, club-shaped, and branching NTs develops in dendrites.
Argyrophilic particles within the cytoplasm tend, initially, to lie loosely dispersed within the deposits of lipofuscin or neuromelanin granules. The central portion of larger NFTs often forms a network around the pigment granules, and these granules may function as an initiation site for promoting further aggregation (Fig. 7.2d, e). This idea is supported by the observation that neither pretangle nor argyrophilic material are found near the Nissl substance. In addition, nerve cell types that do not produce lipofuscin or neuromelanin granules are remarkably resistant to the formation of argyrophilic NTs/NFTs (see also Sect. 9.2).
Mature NFTs are frequently flame-like or comet-like in appearance (Figs. 7.2f and 9.4e, f), whereas other NFT types are rounded or globose. Gradually, NFTs fill large portions of the soma. They even can displace the nucleus toward the periphery and they frequently protrude somewhat into the proximal stems of dendrites; however, they do not extend into the axon (Fig. 7.2d–f).
In persons of advanced age, neocortical layers IIIa and IIIb often contain pyramidal cells that have a massive spindle-shaped enlargement between basal portions of their cell body and the displaced axonal initial segment first beginning at the tip of the enlargement (meganeurites) (Purpura and Baker 1978; Braak 1979). Not only the somatodendritic compartments of involved cells but also the spindle-shaped meganeurites at these locations fill up with AT8-ir or argyrophilic material, thereby confirming the notion that argyrophilic material does not go beyond the axon initial segment into the axon.
NFT Stage I:
Mild argyrophilic lesions develop in the transentorhinal region, involvement of the spinal cord and olfactory bulb
By NFT stage I, all of the non-thalamic nuclei with diffuse cortical projections have developed at least some degree of tau pathology. Along with the cholinergic projection cells of the magnocellular nuclei of the basal forebrain, one also finds AT8-ir material in nerve cells of the hypothalamic tuberomamillary nucleus. At this point, the pathological process also reaches regions of the non-thalamic nuclei that generate descending projections to the lower brainstem and spinal cord, i.e., the lower raphe nuclei and the subcoeruleus nucleus. Axonal networks containing tau aggregates also develop in regions targeted by these nuclei, including the spinal cord, where an AT8-ir axonal network and, more infrequently AT8-ir cell somata are present, decreasing in extent from the cervical to sacral segments (Fig. 9.6a) (Dugger et al. 2013).
Within the cerebral cortex, the transentorhinal region is usually the first site where NTs/NFTs in pyramidal neurons are present (Figs. 7.3a and 9.1a–c). At the same time, one always encounters projection cells there that only show pretangle material. The entorhinal region proper initially remains uninvolved or minimally involved, so that the focus of the tau pathology is clearly in the transentorhinal region. Later in stage I, abundant AT8-ir and Gallyas-positive neurons mark the descent of the superficial entorhinal cellular layer pre-α from its outermost position at the entorhinal border to its deepest position at the transition towards the adjoining temporal neocortex (Fig. 7.3b).
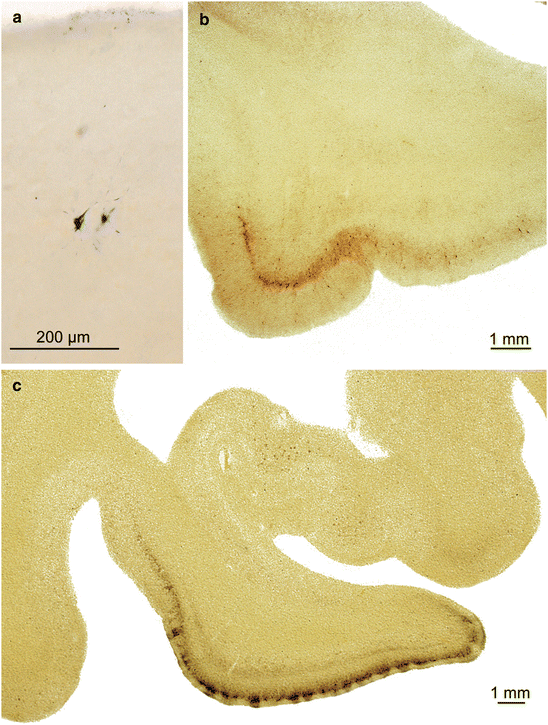

Fig. 7.3
NFT stages I and II in 100 μm sections. (a) Stage I: The transentorhinal region with initial cortical pathology, here in two Gallyas-positive cells. Such subtle lesions can easily be overlooked (49-year-old female, Gallyas silver-iodide impregnation). (b) Advanced NFT stage I: Very large numbers of altered layer pre-α pyramidal cells are seen chiefly in the transentorhinal region, and only a minority of them extend into the entorhinal region proper (AT8 immunoreaction). (c) NFT stage II: Here, not only the transentorhinal region (note the oblique course of the pre-α layer) but also the entire entorhinal region up to the ambient gyrus has become involved. This section also shows the beginning involvement of the deep layer pri-α and subtle involvement of uncal portions of the hippocampal formation (Gallyas silver-iodide impregnation)
Tau pathology occurs at this stage in the anterior olfactory nucleus as well as in tufted and mitral cells of the olfactory bulb, and it increases with disease progression (Fig. 9.6d–f). In subsequent stages, the superordinate cortical components of the olfactory system, including the olfactory portions of the amygdala (cortical subnuclei) and of the entorhinal region (ambient gyrus) become involved. Inasmuch as Aβ deposition in the olfactory bulb is first present in stage III (Attems and Jellinger 2006), the primary pathology there during AD is tangle formation (Kovács et al. 1999).
NFT Stage II:
Argyrophilic lesions progress into the entorhinal region
From the transentorhinal cortex, Gallyas-positive lesions encroach upon the entorhinal region and particularly the superficial cellular layer pre-α (Figs. 7.3c and 9.1d–f). Subsequently, silver-stained lesions also develop in the deep pri-α layer. Both layers gradually become macroscopically visible in thick sections (Fig. 7.3c). Layer pri-α shows sharply defined upper and lower boundaries and it is separated from external layers and sometimes also from the following deep layer, pri-β, by the broad, wedge-shaped lamina dissecans, a myelinated fiber plexus (Fig. 7.3c) (Braak and Braak 1992a; Insausti and Amaral 2012). The presubicular region is uninvolved at this stage. In the hippocampal formation, AT8-ir pyramidal cells begin to appear either in CA 1 or in CA 2. Varicosities filled with abnormal tau material are transiently observable in apical dendrites that pass through the stratum lacunosum moleculare of CA 1 (Braak and Braak 1997a) and lie directly along the way of the perforant pathway (Fig. 9.5a, b). Filigrane networks of AT8-ir axons and dendrites develop in the stratum radiatum and stratum oriens (Fig. 9.5a, b).
7.3 Prevalence of Stages a–II
The relationships among age and AD-associated tau pathology can be studied by staging relatively large numbers of non-selected autopsy cases (Braak et al. 2011). The columns in Fig. 7.4 show the prevalence in n = 1,895/2,366 non-selected cases ranging in age from 1 to 100 years and ranging from those with no tau pathology to those at stages a–II (Table 7.1; Fig. 7.4). The columns represent decades, and the number of cases in each decade that reached the stage(s) shown in each of the four graphs is indicated above the columns (Fig. 7.4a–d). The prevalence of the lesions depicted in Fig. 7.4 does not represent standard epidemiological data because the data were not collected from a living population. Their interpretation rests upon the assumption that the pathological process in AD progresses from stage a to stage VI as a continuum.
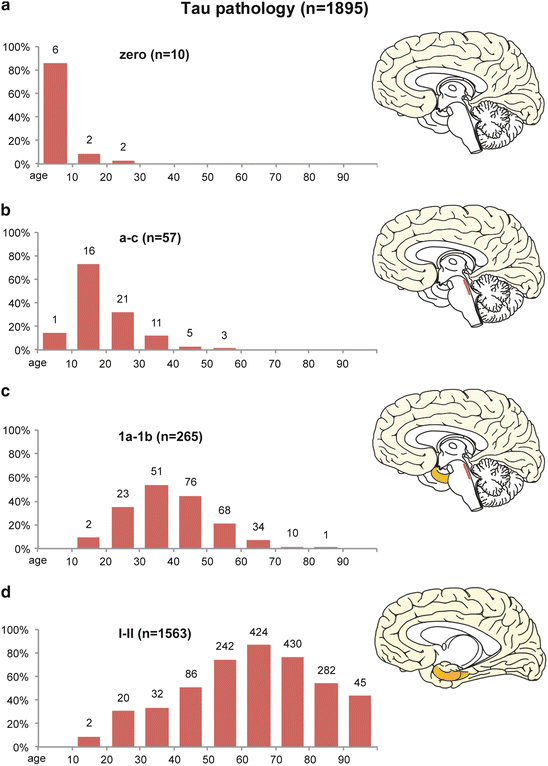

Fig. 7.4
Development of early abnormal intraneuronal tau deposits in n = 1,895 of n = 2,366 non-selected autopsy cases according to decades (ages of the cohort 1–100). Columns display the frequency of cases in relation to the total number of cases in the various age categories. (a) The first row displays the prevalence of cases lacking AD-associated tau aggregates. (b–d) These rows show the evolution of the intraneuronal tau changes: non-fibrillar AT8-ir and still non-argyrophilic aggregates in subcortical stages a–c (b), as opposed to AT8-ir, still non-argyrophilic, aggregates in the cerebral cortex in stages 1a and 1b (c). (d) Argyrophilic neurofibrillary lesions in cortical nerve cells are characteristic of NFT stages I and II
Table 7.1
Development of early tau pathology in n = 1,885 of a total of n = 2,366 cases according to decades, including ratio between females (n = 774) and males (n = 1,111)
Age (n) | Zero (AT8) | a–c (AT8) | 1a–1b (AT8) | NFT I (Gallyas) | NFT II (Gallyas) |
|---|---|---|---|---|---|
0–9 n = 7 | 6 (2/4) 85.71 % | 1 (0/1) 14.28 % | 0 0 % | 0 0 % | 0 0 % |
10–19 n = 22 | 2 (0/2) 9.09 % | 16 (3/13) 72.72 % | 2 (0/2) 9.09 % | 2 (0/2) 9.09 % | 0 0 % |
20–29 n = 66 | 2 (2/0) 3.03 % | 21 (9/12) 31.81 % | 23 (9/14) 34.84 % | 18 (8/10) 27.27 % | 2 (2/0) 3.03 % |
30–39 n = 95 | 0 0 % | 11 (3/8) 11.58 % | 51 (28/23) 53.68 % | 28 (12/16) 29.47 % | 4 (0/4) 4.21 % |
40–49 n = 170 | 0 0 % | 5 (3/2) 2.94 % | 76 (28/48) 44.70 % | 81 (35/46) 47.65 % | 5 (3/2) 2.94 % |
50–59 n = 326 | 0 0 % | 3 (0/3) 0.92 % | 68 (28/40) 20.86 % | 219 (72/147) 67.18 % | 23 (1/22) 7.06 % |
60–69 n = 487 | 0 0 % | 0 0 % | 34 (9/25) 6.98 % | 305 (107/198) 62.63 % | 119 (42/77) 24.44 % |
70–79 n = 564 | 0 0 % | 0 0 % | 10 (4/6) 1.77 % | 222 (93/129) 39.36 % | 208 (100/108) 36.88 % |
80–89 n = 525 | 0 0 % | 0 0 % | 1 (0/1) 0.19 % | 94 (48/46) 17.90 % | 188 (101/87) 35.81 % |
90–100 n = 104 | 0 0 % | 0 0 % | 0 0 % | 11 (6/5) 10.58 % | 34 (20/14) 32.69 % |
Total n = 2366 | 10 (4/6) 0.42 % | 57 (18/39) 2.41 % | 265 (106/159) 11.20 % | 980 (381/599) 41.42 % | 583 (269/314) 24.64 % |
Remarkably, only a few cases show the complete absence of abnormal tau inclusions in the CNS (10/2,366 cases = less than 1 % of all cases) (Table 7.1; Fig. 7.4a). Six of these ten cases are younger than 10 years of age and all of them are devoid of Aβ deposition (Table 7.1). The inclusion of ‘negative’ results here and below is not unproblematic: In contrast to a positive finding, the validity of a negative finding is subject to limitations, e.g., here, assessment of a single 100 μm tissue section for each region. Where a negative finding results, it cannot be ruled out that, under other circumstances or conditions (control of more sections), the outcome may have been different.
As seen in Fig. 7.4b–d, more than 99 % (and, as of the fourth decade, all) of the individuals sampled show the presence of early tau pathology in one or the other stage. These intraneuronal lesions in the human CNS are not benign or ‘normal’ because they become increasingly severe and cause cellular dysfunction ultimately leading to the premature death of the involved neurons.
Cases in Fig. 7.4b exhibit the presence of AT8-ir material only in the locus coeruleus and/or other nuclei with ascending and diffuse cortical projections (subcortical stages a–c: 57 of all 2,366 cases = approximately 2 %) (Table 7.1; Fig. 7.4b). The cerebral cortex and other brain regions are devoid of AT8-ir material. Cases characterized by the three lesional distribution patterns ‘a’ (tau aggregates in axons of the locus coeruleus), ‘b’ (tau aggregates in melanized neurons of the locus coeruleus), and ‘c’ (tau aggregates in the locus coeruleus and in other subcortical nuclei with diffuse cortical projections) occur, surprisingly, at very young ages. The fact that irreversibly hyperphosphorylated and slightly aggregated tau in axons and pretangle material in the somatodendritic compartment develop in brainstem nuclei of young individuals, implies that advanced age alone is not mandatory for the production of the pathological material. Overview sections stained for lipofuscin pigment and basophilic material do not reveal any obvious pathological alterations (e.g., loss of basophilic material, displacement of cell nuclei to the periphery). The prevalence of such cases culminates in the second decade and slowly decreases thereafter, although these stages are present until the end of the sixth decade (Table 7.1; Fig. 7.3b). The intraneuronal pathology during the early stages a–c is not accompanied by insoluble extracellular deposits of Aβ (Braak et al. 2011; Braak and Del Tredici 2011, 2012).
The brainstem nuclei with diffuse cortical projections differ essentially from other systems within the CNS. As such, it should become possible in the future to detect even incipient or slight disturbances within the noradrenergic and other systems by developing new and sensitive non-invasive tests. Systemic abnormalities potentially caused by very early AD-associated lesions in young persons (for instance, early dysregulation of the intracerebral microvascular system and the blood brain barrier) may have been overlooked or misinterpreted up to now because, from a differential diagnostic standpoint, they may not have been viewed within the context of the AD-process.
A third graph shows a total of 265 cases in which, in addition to subcortical lesions, cortical tau pathology occurs for the first time, above all in the transentorhinal region (stages 1a and 1b) (Table 7.1; Fig. 7.4c). This intraneuronal material is AT8-ir but non-argyrophilic. In stage 1a (38/2,366 cases = approximately 2 %), it is confined solely to neuritic processes, very probably extrinsic axons. Also included in this graphic are cases with cortical AT8-ir pyramidal cells that mostly represent modified layer pre-α cells of the transentorhinal region (stage 1b: 227/2,366 cases = approximately 9 %) (Table 7.1; Fig. 7.4c). Some individuals display only a single involved pyramidal cell, whereas others exhibit more AT8-ir neurons. All 1a and 1b cases (265/2,366 cases = approximately 11 %) have patterns of subcortical lesions resembling those in stages a–c; in other words, the cortical tau lesions do not occur in the absence of subcortical tau pathology. The prevalence of cases in stages 1a and 1b increases steadily up to the fourth decade. Thereafter, it slowly decreases up to the ninth decade (Table 7.1; Fig. 7.4c). Some individuals at stages 1a/1b also develop initial Aβ deposits (12 at phase 1 = approximately 4 % of the 265 1a/1b cases and 9 at phase 2 = approximately 3 % of the 265 1a/1b cases) (Table 7.2). See also Chap. 8.
Table 7.2
Comparison of early stages of AD-associated tau lesions with phases of Aβ deposition (n = 1,885)
Aβ phases | Tau zero | Tau stages a–c | Tau stages 1a–1b | NFT stage I | NFT stage II | Stages a–II |
|---|---|---|---|---|---|---|
Zero | 10 | 57 | 244 | 687 | 265 | 1,253 |
1 | 0 | 0 | 12 | 141 | 103 | 256 |
2 | 0 | 0 | 9 | 128 | 135 | 272 |
3 | 0 | 0 | 0 | 22 | 74 | 96 |
4 | 0 | 0 | 0 | 2 | 6 | 8 |
5
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|




