and Kelly Del Tredici1
(1)
Zentrum f. Biomed. Forschung AG Klinische Neuroanatomie/Abteilung Neurologie, Universität Ulm, Ulm, Germany
6.1 The Cerebral Cortex
The cerebral cortex is the overarching superordinate structure of the human CNS. It is not a uniform entity but consists of two fundamentally different types of gray matter, i.e., the expansive and, for the most part, uniformly composed neocortex (Fig. 6.1a, yellow, light and deep orange, light and deep blue shading) and a small and heterogeneous allocortex (Fig. 6.1a, red shading). A transition region mediates between the two (Fig. 6.1a, green shading) (Braak 1980; Nieuwenhuys 1994; Amunts and Zilles 2001; Zilles and Amunts 2010). The neocortex is chiefly responsible for processing and planning the interactions with the external world. It receives abundant somatosensory, visual, and auditory data, and it influences, at the same time, somatomotor activity that impinges on the organism’s environment (Fig. 6.1c). The neocortex, which takes up approximately 95 % of the total surface area of the human cerebral cortex, generally shows a six-layered organization, with the exception of a few regional variations.
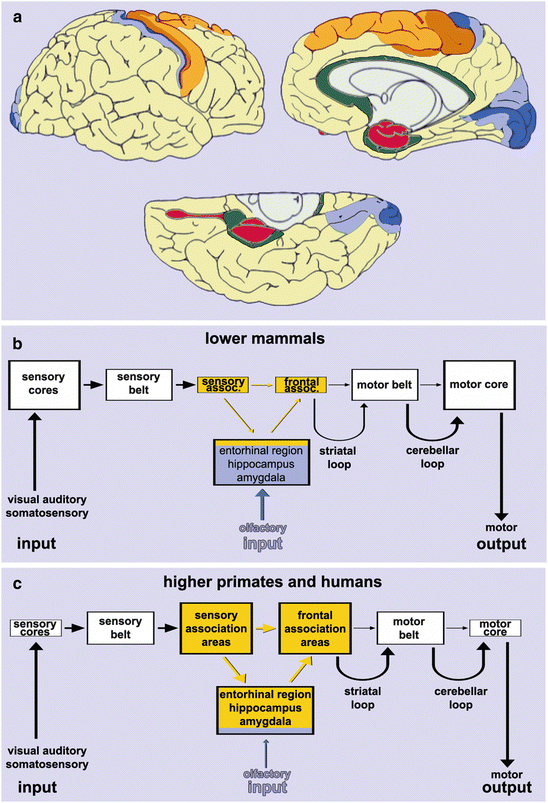

Fig. 6.1
Major subdivisions of the cerebral cortex. (a) The cerebral cortex consists of two basic types of gray matter: an extensive neocortex and a small allocortex (red). Green areas of the periallocortex and proneocortex mediate between the two. The allocortex is located chiefly in anteromedial portions of the temporal lobe. It includes the olfactory bulb and anterior olfactory nucleus, on the one hand, and limbic system centers, such as the hippocampal formation, presubiculum and entorhinal region, on the other. The subcortical amygdala is closely related. The neocortex is remarkably extensive in the human brain. The frontal, parietal, occipital, and temporal lobes each have a primary core area (deep orange and deep blue), a belt of first order association areas (light orange and light blue), and related high order association areas (yellow). (b) Macrosmatic mammals process olfactory input, in part, in the entorhinal region, which has only weak connectivities to the less well developed high order association areas of the neocortex. (c) By contrast, microsmatic primates and humans possess limited olfactory input and utilize the entorhinal region to process essentially neocortical information. The much larger high order association areas are connected to the ‘newly’ organized entorhinal region via the strong afferent and efferent trunks of the limbic circuit
Neocortical areas of the parietal, occipital, and temporal lobes are fundamentally divided into highly refined and maturely myelinated primary fields (core fields) that are responsible for the initial processing of incoming sensory data (Fig. 6.1a, deep blue). Each of the diverse sensory cores is encircled by a zone of somewhat less highly differentiated and only moderately myelinated unimodal first order sensory association areas (Fig. 6.1a, light blue). These first order areas, in turn, are interconnected with extensive but more simply organized and incompletely myelinated unimodal or heteromodal high order sensory association areas (Fig. 6.1a, yellow). The frontal lobe is similarly structured into a primary motor (core) field (Fig. 6.1a, deep orange) and premotor (belt) areas (Fig. 6.1a, light orange), the latter of which give way to the extensive high order frontal association areas, the prefrontal cortex (Fig. 6.1a, yellow). The enormous dimension of the prefrontal cortex, this highest executive instance in the CNS, is a distinct feature of the human brain.
By comparison, the heterogeneously composed allocortex is small (Fig. 6.1a, red). The number of layers varies from one region to another, ranging from simpler trilaminate structures (e.g., dentate fascia) to more complex structures with more than six layers (e.g., entorhinal region). The mammalian brain contains two allocortical cores: first, the olfactory bulb and areas related to it and, second, the hippocampal formation, presubiculum, and entorhinal region (Braak and Braak 1992a; Braak et al. 1996). In the microsmatic human brain, most structures devoted to olfactory processing are rudimentary (Fig. 6.1c) in comparison to the corresponding areas in macrosmatic mammals (Fig. 6.1b), whereas the hippocampal formation and regions related to it are highly developed. The hippocampus lacks direct sensory input and must rely, instead, on indirect input from both olfactory structures and the neocortex to obtain information about the world outside the individual (Fig. 6.1c).
Transition zones between the allocortex and neocortex include a zone of periallocortical areas allied with the allocortex in the narrowest sense and proneocortical areas leading to the mature neocortex (Fig. 6.1a, green). Because allocortical layers can transgress the limits of their own territories, the periallocortex often appears as an admixture of both allocortical and neocortical layers, whereas the proneocortex is usually dysgranular in character with poorly developed layers II and IV (Braak 1980).
6.2 The Amygdala
The amygdala is located in the anteromedial portion of the temporal lobe directly anterior to the hippocampal formation (Fig. 6.2). It chiefly consists of medial, cortical, and central subnuclei, supplemented by a voluminous basolateral complex that includes lateral, basal, and accessory basal subnuclei (Fig. 6.2) (Alheid 2003; Yilmazer-Hanke 2012; Bzdok et al. 2013). The amygdala is bilaterally and closely interconnected with cortical components of the limbic system: the entorhinal region, presubiculum, and hippocampal formation. Projections from olfactory areas terminate preferentially in cortical subnuclei, whereas the central subnucleus receives crucial interoceptive input from internal organs and regulates not only endocrine and autonomic functions but also learning, memory, and emotional reactions. Moreover, the central subnucleus controls all of the non-thalamic nuclei that generate diffuse projections to the cerebral cortex (Bohus et al. 1996; Nieuwenhuys 1996). The nuclei of the basolateral complex have bidirectional interconnectivities with neocortical high order association areas (Fig. 6.2). The lateral subnucleus is the major entry point for neocortical information. Dense projections from the basal and accessory basal subnuclei feed into the efferent trunk of the limbic circuit, which, in the human brain, heavily influences the prefrontal cortex via the ventral striatum, ventral pallidum, and dorsomedial nuclei of the thalamus (Fig. 6.10) (Heimer et al. 1991; Alheid 2003; Haber and Gdowski 2004).
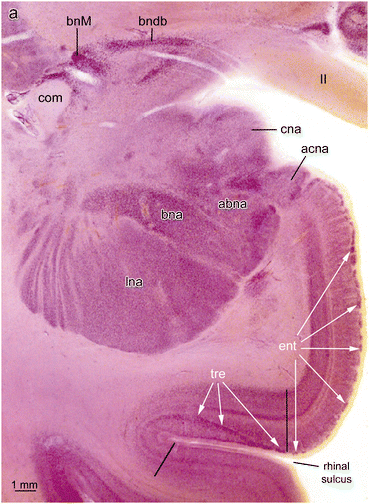

Fig. 6.2
Pigment-Nissl stained 200 μm section of the human entorhinal region cut at the latitude of amygdala. The entorhinal region (ent) covers almost totally the free surface of the parahippocampal gyrus, whereas the transentorhinal region (tre) is concealed deep in the rhinal sulcus. The intensely staining islands of pre-α (white arrows) coalesce in the vicinity of the rhinal sulcus and descend obliquely through the outer layers of the transentorhinal region the borders of which are indicated by black lines. The amygdala at this latitude shows cortical (cna) and accessory cortical subnuclei (acna). The large basolateral complex consists of three subnuclei, the accessory basal subnucleus (abna), basal subnucleus (bna), and lateral subnucleus (lna). Abbreviations: acna—accessory cortical subnucleus of the amygdala; bndb—bed nucleus of the diagonal band; bnM—basal nucleus of Meynert; cna—cortical subnucleus of the amygdala; com—anterior commissure; II—optic tract
6.3 The Entorhinal Region and the Presubiculum
The entorhinal region is the only association cortex of the hippocampal formation and extends over the ambient gyrus and anterior portions of the parahippocampal gyrus where small wartlike elevations (verrucae hippocampi) separated by shallow grooves mark the position of the entorhinal region and are visible to the naked eye (Fig. 6.4a) (Augustinack et al. 2012). The borders of the entorhinal cortex often coincide with the course of the rhinal sulcus anteriorly and the collateral sulcus posteriorly (Fig. 6.4a) (Hanke 1997). Presently, it is unknown whether and to what an extent variability of the entorhinal region reflects individual differences in personality or intellectural capacity (Amunts et al. 2005).
Notably, none of the entorhinal layers corresponds to a layer of the neocortex. To avoid confusion, a terminology distinguishing between external and internal principal strata (pre versus pri) is used throughout the text here (Figs. 6.3 and 9.1) (Braak and Braak 1992a). Large multipolar projection cells of layer pre-α are bunched together into characteristic cellular islands (Fig. 6.3), which occur nowhere else in the cerebral cortex. The external layers, pre-α, pre-β, and pre-γ give rise to the perforant pathway, which crosses the obliterated hippocampal fissure and terminates in the outer two-thirds of the dentate molecular layer, subiculum, and first sector of the Ammon’s horn (CA 1) (Fig. 6.4c) (Witter 1993).
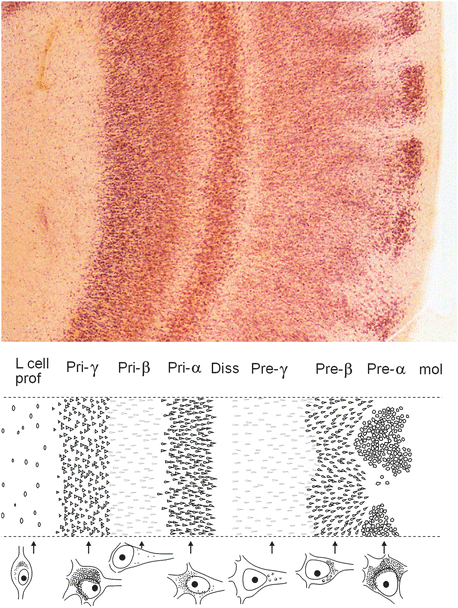
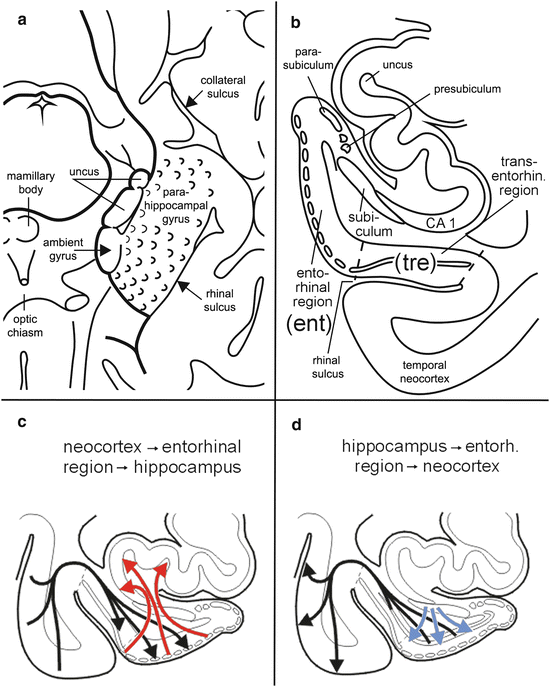

Fig. 6.3
Pigment-Nissl stained 200 μm section of the human entorhinal cortex and a detailed scheme of lamination as well as characteristic nerve cells types in nine layers based on pigmentation (lipofuscin granule) patterns. Drawing shows the cellular layers located in the outer and inner main strata (Pre and Pri). Abbreviations: mol—molecular layer; Pre-α—layer pre-α, Pre-β—layer pre-β, Pre-γ—layer pre-γ (cellular layers of the outer main stratum—external principal stratum); Diss—Lamina dissecans; Pri-α—layer pri-α, Pri-β—layer pri-β, Pri-γ—layer pri-γ (cellular layers of the inner main stratum—internal principal stratum); L cell prof—Lamina cellularis profunda. Diagram adapted with permission from H Braak and E Braak, Temporal sequence of Alzheimer’s disease-related pathology. Cerebral Cortex 1999;14:475–512

Fig. 6.4
Macroscopic topography of the human entorhinal cortex and schemata of major entorhinal region afferent and efferent projections. (a) Diagram showing the topographical relationships of the entorhinal region, which extends over anteromedial portions of the temporal lobe. The surface of the entorhinal cortex (parahippocampal gyrus) exhibits wart-like elevations, the verrucae hippocampi. Note the uncus of the hippocampus, which reaches the free surface of the brain. (b) Frontal section through the uncal portion of the hippocampus, the adjoining parahippocampal gyrus and occipitotemporal gyrus. (c, d) Diagrams of the major entorhinal connectivities. (c) Neocortical input (black arrows) together with input from limbic circuits. The borders of the entorhinal and transentorhinal regions are marked by a dashed line. Converges upon the transentorhinal region and layer pre-α of the entorhinal region. From there, it is transferred through the perforant pathway (red arrows) heading to the hippocampal formation. (d) A dense back-projection from the subiculum of the hippocampal formation terminates in the entorhinal deep layer pri-α (red arrows) and is then transferred back to the neocortex (black arrows). Thus, the entorhinal and transentorhinal regions serve predominantly as an interface between the neocortex and hippocampal formation. Abbreviations: CA 1—cornu Ammonis (first sector of the Ammon’s horn); ent—entorhinal region; tre—transentorhinal region. Micrographs (a) and (b) are adapted with permission from H Braak et al., Acta Neuropathol 2006;112:389–404
The presubiculum consists of cloud-like accumulations of small projection neurons supplemented by deep layers of the hippocampal subiculum, whereas the adjacent parasubiculum includes small neurons interfacing with a deep layer of the entorhinal region (Fig. 6.4b) (Kalus et al. 1989). Various versions of pathways subsumed under the name ‘Papez circuit’ arrive at the entorhinal region via the presubicular parvocellular layer (Fig. 6.10).
The transentorhinal region is situated along the lateral circumference of the entorhinal region deep in the rhinal sulcus (Figs. 6.2 and 6.4a, b) (Taylor and Probst 2008; Ding and van Hoesen 2010). The region is largest in the human brain and decreases markedly in size further down the primate scale (Braak and Braak 1992a). The transentorhinal region is easily identifiable by the gradual oblique descent of layer pre-α through the outer main stratum of the cortex (Fig. 6.2). Owing to this distinctive attribute, the transentorhinal region is one of the best characterized areas of the human cerebral cortex.
Together, the transentorhinal and entorhinal regions in higher primates and humans function as an interface between the neocortex and the hippocampal formation (Fig. 6.4c, d). Since virtually all of the data exchanged between the neocortex and hippocampus must pass through both regions (or else through the lateral subnucleus of the amygdala), bilateral damage to the transentorhinal and entorhinal regions and the amygdala impairs this dataflow and ultimately interferes with higher cortical capabilities (Fig. 6.10).
Given the unique status of the entorhinal region in the human brain, the region is shown here in sections cut tangentially to the cortical surface, so as to display the patterns of the approximately 250 cellular islands in the superficial layer pre-α (Fig. 6.5a–d). The pattern of each individual is unique (compare Fig. 6.5c with d): in some cases, the islands are large but few (Fig. 6.5d), in others numerous small islands are present (Fig. 6.5c). In some individuals, a large proportion of the cellular islands in the pre-α layer have a reticulate pattern, in others they are detached. The right and left entorhinal regions of each person are similar, like the fingerprints of both hands (Fig. 6.5a right, b left). Remarkably, the entorhinal cortex—and, above all, its superficial layer of cellular islands, which transfers crucial information between the neocortex and the hippocampal formation—is highly susceptible to early-appearing and severe pathological changes in the AD process (Braak and Braak 1991a, 1999).
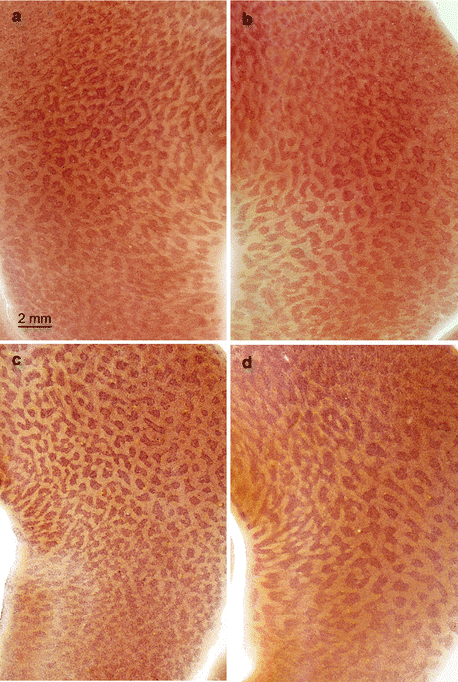

Fig. 6.5
Pigment-Nissl stained 200 μm sections cut tangentially to the surface of the entorhinal cortex from individual brains. In all sections, the highly unusual cellular islands of layer pre-α are visible. Whereas the patterns in layer pre-α of the left and right entorhinal cortex of a single individual resemble each other very closely (a, b), the unique patterns of different individuals make it possible to distinguish these from each other (c, d). Reproduced with permission from H Braak and E Braak, Staging of Alzheimer’s disease-related neurofibrillary changes. Neurobiol Aging 1995;16:271–284. Scale bar in (a) also applies to micrographs in (b–d)
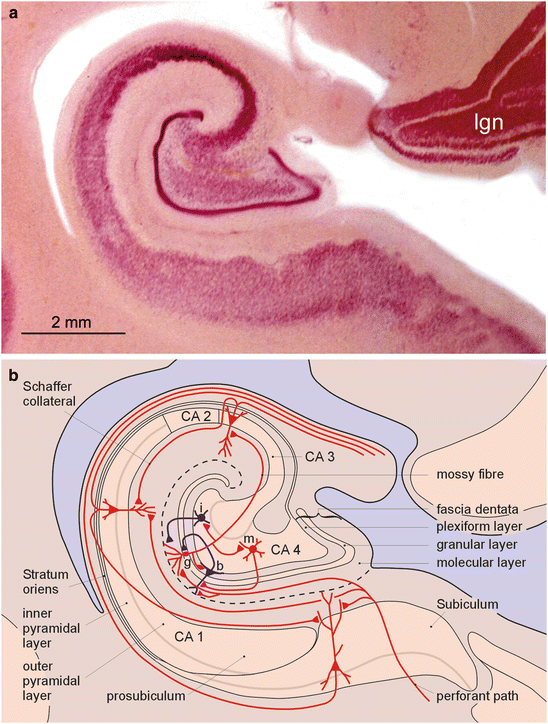
6.4 The Hippocampal Formation
The hippocampus is almost completely hidden in the depths of the temporal lobe and gradually decreases in size en-route to the splenium of the corpus callosum. Three major units can be distinguished: the dentate fascia, the Ammon’s horn (cornu Ammonis) with its four sectors (CA 1–CA 4), and the subiculum (Fig. 6.6). In the neocortex, layers that preferentially receive afferent fibers and those generating efferent projections are arranged one on top of the other. However, in the hippocampal formation, the layers lie side by side. The dentate fascia is a kind of hippocampal granular cortex that is specialized at receiving a number of different afferents. It consists chiefly of small projection cells that functionally correspond to the granule cells of the fourth neocortical layer (spiny stellate cells). Both the Ammon’s horn and the subiculum, by contrast, are dominated by large pyramidal cells that furnish the output pathways and functionally correspond to neocortical layers III and V (Braak et al. 1996; Gloor 1997; Insausti and Amaral 2012).