Diagnostic Endovenous Procedures
While endovascular therapies are normally associated with the treatment of vascular disease, there are a myriad of procedures that use endovascular techniques to aid in the diagnosis of various conditions. Some of the more common endovascular diagnostic procedures are discussed below.
Inferior Petrosal Sinus Sampling
Etiology
In the early 1980s, Oldfield and Doppman from the National Institutes of Health introduced bilateral inferior petrosal sinus sampling (IPSS), which is used by both neurosurgeons and endocrinologists to determine whether the potential source of aberrant hormone secretions, such as adrenocorticotropic hormone (ACTH), is the pituitary gland. This diagnostic test is indicated when imaging studies do not demonstrate an obvious pituitary mass that could be considered responsible for hormonal imbalances, such as those seen in Cushing syndrome. When no overt pituitary mass is observed, an ectopic source of ACTH secretion is often suspected, but it is challenging to differentiate between an ectopic source of ACTH and a pituitary source of ACTH clinically or using only biochemical tests. Bilateral IPSS is used to differentiate whether the ACTH causing Cushing syndrome comes from the pituitary or is ectopic in origin. It may also help localize which side of the gland is producing ACTH at abnormally high levels.
Cushing syndrome is a clinical condition that results after prolonged exposure to ACTH. Of the cases in which hypercortisolemia results from an ACTH–dependent process, approximately 80% are due to a pituitary adenoma (Cushing disease), 10% are due to adrenal lesions, and the remaining 10% are secondary to ectopic ACTH secretion. Large pituitary adenomas (>1 cm diameter) are rare and usually present with mild manifestations of Cushing’s syndrome. Pituitary microadenomas (<1 cm diameter) are mainly responsible for ACTH-dependent Cushing syndrome. However, precise localization of these microadenomas is often difficult, because of their small size. Surgical removal of tumors secreting ACTH is the treatment of choice in Cushing disease, which makes finding the source of ACTH secretion paramount in the management of Cushing disease.
Diagnosis
The clinical features of a pituitary adenoma vary depending on the location, size of the tumor, and its secretory function and capability. Pituitary macroadenomas can cause headaches, double vision, loss of peripheral vision leading to blindness, facial pain, or numbness. Hypopituitarism manifests itself by lack of energy, weight loss, nausea, vomiting, constipation, amenorrhea and infertility, dry skin, increased pigmentation of the skin, cold intolerance, and mental status changes. ACTH-secreting adenomas produce Cushing disease, which itself results in moon facies, with acne and flushing, fatty deposits over the back of the neck, stretch marks, easy bruising, posterior subcapsular cataracts, arterial hypertension, aberrant hair growth, diabetes mellitus, muscle loss, fatigue, depression, and psychosis.
Visual acuity can be decreased in one or both eyes. Bitemporal superior quadrantanopsia can be seen in patients with a macroadenoma causing compression of the optic chiasm; larger adenomas can cause bitemporal hemianopsia. Optic atrophy is frequently seen on a fundus exam. Galactorrhea can be seen in patients with prolactinomas. In cases of acromegaly, which is due to excess growth hormone, patients can present with large hands and feet, coarse facial features with frontal bossing, high arched palate, and prognathism. If the disease is left untreated, patients will usually succumb to these chronic conditions.
Conventional imaging studies like CT and MRI have relatively poor sensitivity in identifying pituitary lesions.1–3 Dynamic MRI, in which the parasellar region is imaged continuously before and after contrast administration, apparently has higher sensitivity with lower specificity for localizing microadenomas. Studies have shown that MRI has a range of sensitivity from 46% to 67% in detecting ACTH-secreting pituitary microadenoma and a range of specificity from 62% to 100%.1,4,5 However, these imaging studies have inherent diagnostic limitations, given the potential significant incidence of asymptomatic microadenomas and difficulty in locating small ectopic sites.
Evidence-Based Medicine
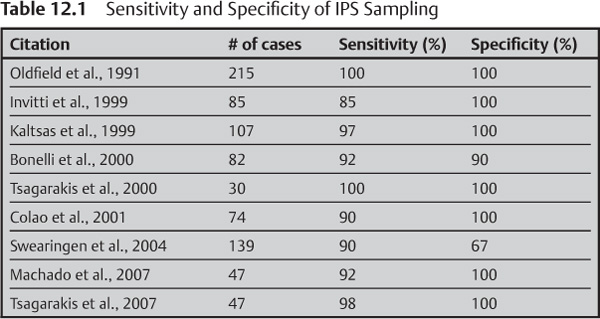
Sensitivity and Specificity of Inferior Petrosal Sinus Sampling
Inferior petrosal sinus sampling is considered the gold standard for differentiating whether the source of ACTH secretion in patients with Cushing disease is ectopic of from the putuitary.6–9 The sensitivity of inferior petrosal sampling ranges from 85% to 100% and the specificity ranges from 67% to 100% (Table 12.1). A hypo-plastic or anomalous inferior petrosal sinus was believed to underlie the false-negative results of inferior petrosal sinus sampling. Other causes of ambiguous results include IPSS performed during a period of normal cortisol levels in patients with intermittent ectopic ACTH secretion and false-positive test results caused by CRH-secreting tumors.10 During inferior petrosal sinus sampling, diagnostic accuracy can be increased by additional sampling of other anterior pituitary hormones, such as prolactin, for normalization of ACTH ratios. Booth et al. compared the efficacy of IPSS and the results of imaging studies for localization of pituitary tumors and demonstrated a 70% likelihood of accurate localization using inferior petrosal sinus sampling compared with 49% using imaging alone.11–13 Selective venous sampling from the cavernous sinus is an alternative to inferior petrosal sinus sampling and is highly reliable in lateralizing the tumor location.
Preprocedural Preparation
Inferior petrosal sinus sampling is used for both localization and differential diagnosis in the face of hormonal imbalances and inconclusive imaging studies. Patients selected for IPSS usually have a sustained elevation of free cortisol in their urine (>250 µg/day) and are not on any suppressive drugs. If they are on suppressive medications, the effect of such drugs should be allowed to dissipate completely for some weeks prior to IPSS. Patients who have had a bilateral adrenalectomy are not suitable candidates for IPSS. In these cases, dexamethasone 0.5 mg every 6 hours for 2 to 10 days prior to IPSS suppresses the normal pituitary corticotrophs. As with other angiographic procedures, NPO status should be enforced the night before, and standard moderate sedation protocols can be used. The patient should also be asked about any history of a deep venous thrombus or IVC filter placement, as this may alter the route of access. Patients should always have routine laboratory tests performed and should be asked about dye allergies and medications that may interfere with the use of contrast materials.
Equipment
Fifteen lavender-top tubes with EDTA should be labeled, numbered, and placed in an ice-water bath prior to beginning IPSS. During the procedure, each sample is drawn into a syringe and transferred into the appropriately labeled lavender-top tube. Samples should be sent to the laboratory immediately for processing, including centrifugation within 1 hour of collection.
Sheath and Catheter
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree