EPIDEMIOLOGY
In 2013, chronic liver disease was the 12th leading cause of death in the United States accounting for more than 36,000 deaths and over 100,000 hospitalizations. Those figures are probably an underestimate and the prevalence is steadily increasing. Hepatitis C patients, typically born between 1945 and 1965, are contributing to a large number of new cases and the obesity epidemic accounts for an increase in nonalcoholic fatty liver disease.
The less severe form of neurologic impairment, minimal hepatic encephalopathy (MHE), afflicts between 60% and 80% of cirrhotic patients. Often qualified of “subclinical,” MHE is nonetheless a tremendous social burden with the majority of patients being unfit to drive and half of them unable to keep a permanent job.
Overt hepatic encephalopathy occurs in 30% to 45% of cirrhotic patients. It is responsible for the bulk of hospitalization days attributed to cirrhosis and its complications. Depending on the time course, it is subdivided into episodic, recurrent (6 months or less between episodes), or persistent (behavioral alterations that are always present).
PATHOBIOLOGY
Causes of chronic liver damage are numerous (
Table 119.2), and the final common pathway is cirrhosis defined by parenchymal fibrosis with hepatic architecture distortion and the formation of regenerative nodules. The numerous associated complications are related to a loss of synthetic and metabolic functions, relative immunosuppression, and portal hypertension often associated to portosystemic shunting.
Most of the existing evidence points to ammonia (NH3) as the main culprit behind HE. It is generated from glutamine by the enterocytes and from catabolism of nitrogenous products (proteins and urea) by colonic bacteria. Detoxification normally takes place in the liver through the conversion of NH3 to urea and glutamine basically reversing what took place in the digestive tract. Cirrhosis and portosystemic shunting bypasses this process, allowing systemic levels of NH3 to rise, which easily crosses the blood-brain barrier by diffusion and ion channels. Once in the brain, NH3 is taken up by astrocytes, where it triggers oxidative stress, increases intracellular calcium, induces mitochondrial dysfunction, and activates NF-kB and an inflammatory response. Some data suggest that severity of HE correlates more with inflammatory indices than with NH3 levels. Many other molecules have been implicated in the pathobiology of HE, such as neurosteroids. Synthetized by astrocytes and microglia, they appear to upmodulate the α-aminobutyric acid (GABA)-A receptors leading to the clinically obvious increased GABA-ergic tone (depression of central nervous system [CNS] function) seen in HE.
CLINICAL FEATURES
Cirrhosis
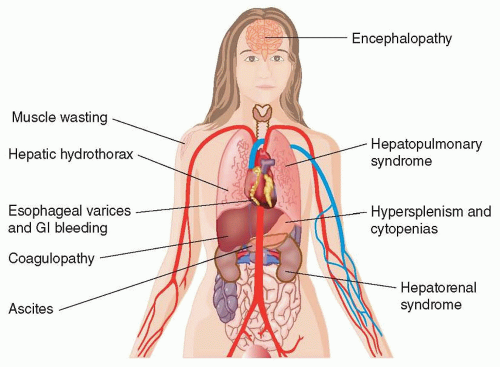
Compensated cirrhotic patients may be asymptomatic, but eventually, most will develop one of the myriad of possible clinical manifestations as the disease progresses (
Fig. 119.1). They may report nonspecific symptoms like anorexia, weight loss, fatigue, and weakness. They often have a low blood pressure, and they may present jaundice, pruritus, severe muscle cramps, or abdominal distention from ascites.
Classically, cirrhotic patients are classified according to the Child-Turcotte-Pugh score. Designed 30 years ago to predict outcome after surgery for portal hypertension (
Table 119.3) (http://www.mdcalc.com/child-pugh-score-for-cirrhosis-mortality), it is now used to grade clinical severity and follow the evolution of most patients. Alternatively, the Model for End-Stage Liver Disease (MELD) score evaluates serum creatinine, bilirubin, and international normalized ratio (INR) to predict 3-month mortality following transjugular intrahepatic portosystemic shunt (TIPS) procedures (http://www.mdcalc.com/meld-score-model-for-endstage-liver-disease-12-and-older/). The MELD score is currently used to prioritize candidates for liver transplantation scaled from 6 (less ill) to 40 (gravely ill).
Minimal Hepatic Encephalopathy
Patients with MHE, by definition, have no clinically detectable abnormalities at the bedside but manifest clear deficits on psychometric testing. They usually reveal impairment of vigilance, alertness and orientation, executive functions, working memory, learning processes, visuospatial coordination, and reaction time. When looked at closely, daily functioning is usually impaired in all spheres. They have a higher risk of car accidents with less than 20% of them fit to drive. Verbal ability is characteristically unimpaired.
Overt Hepatic Encephalopathy
Overt hepatic encephalopathy (OHE) can manifest itself from subtle clinical signs to profound coma. As it sets in, one of the earliest manifestations is usually a change in the sleep pattern (usually hypersomnia). Once it progresses, behavioral changes such as apathy, anxiety, and irritability appear. Attention span diminishes and short-term memory becomes impaired. Patients sometimes suffer from visual perception disturbances, both as a result of cortical and retinal dysfunction, with visual agnosia, macropsia, spatial disorientation, and problems with visuospatial construction. Visual hallucinations can occur. Neuromuscular impairments include ataxia, dysarthria, hyperreflexia, and extensor plantar responses. Asterixis, although not pathognomonic, was first described in HE patients. This “flapping tremor” is a negative myoclonus characterized by brief interruptions of sustained voluntary muscle contractions causing loss of postural tone. It is usually bilateral, nonrhythmic, and with a frequency of 3 to 5 Hz. It can be elicited by hyperextension of the wrists but is also seen in the feet, tongue, or eyelids. Parkinsonian features are at times prominent with tremor, rigidity, bradykinesia, hypomimia, slowness of speech, and monotony. Focal deficits have been described but are rare. As patients deteriorate, the most striking feature is the mental status change from lethargy to stupor and coma. At the point of coma, most patients are hyperventilating and have loss of deep tendon reflexes. Seizures are unusual. Mental status and motor changes do not always progress in a synchronized fashion.
Hepatic Myelopathy
Another well-described long-term neurologic complication of cirrhosis is hepatic myelopathy (HM). It is characterized by severe spastic paraparesis with minimal sensory involvement. Spasticity
and weakness usually affect the lower limbs more severely and are progressive. It is caused by symmetric demyelination of the lateral pyramidal tracts. It does not respond to NH
3-lowering therapy, although some improvement might follow liver transplantation.
Persistent Hepatic Encephalopathy
Often preceded by multiple episodes of OHE, a chronic and largely irreversible syndrome characterized by dementia, dysarthria, gait ataxia, intention tremor, and choreoathetosis might appear. This persistent form of HE, previously referred to as acquired hepatolenticular degeneration, afflicts 4% of cirrhotic patients. The associated neuropathologic findings are neuronal necrosis in basal ganglia, cerebellum, and cerebral cortex in addition to Alzheimer type II astrocytosis. It does not respond to standard therapies.