45 Management of Vascular Complications During Endoscopic Skull Base Surgery
Ricardo L. Carrau, Juan C. Fernandez-Miranda, Daniel M. Prevedello, Paul A. Gardner, Carl H. Snyderman, and Amin B. Kassam
 Introduction
Introduction
Prevention and management of complications is an important cornerstone of surgery and directly relates to the first tenet of “do no harm.” As different surgical specialties evolved into minimally invasive approaches, using new technique or technologies, they universally encountered a higher frequency of surgical complications. Even the most experienced surgeons are not immune to complications, but they can be ameliorated by proper training and mentorship that promotes and facilitates sequential and progressive learning. Despite its minimally invasive approach, endoscopic skull base surgery presents many of the risks and the potential for major complications that are associated with traditional approaches. Inexperience and lack of proper training with new instruments or techniques, and disorientation with the endoscopic anatomical perspective are important factors leading to catastrophic vascular injuries.
In our experience (which includes over 1400 endoscopic endonasal skull base surgeries), the incidence and morbidity of catastrophic vascular complications compare favorably to that of traditional approaches. However, in our practice endoscopic and traditional approaches are complementary, and therefore their indications and limitations preclude a direct comparison. This chapter summarizes our experience and presents guidelines for the prevention and management of vascular complications during endoscopic skull base surgery.
 Incidence of Complications
Incidence of Complications
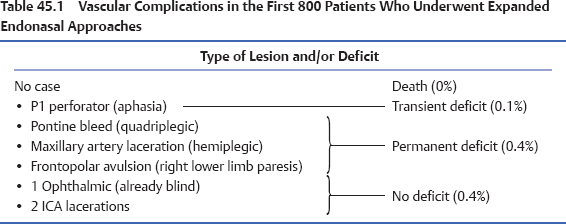
Vascular complications may be classified by the type of vessel (capillary, venous, or arterial), size of the vessel (minor or major), flow of the vessel (low flow or high flow), severity of the complication (major or minor), location (intra- or extracranial), and timing (intraoperative or postoperative). Table 45.1 summarizes the vascular complications of our first 800 endoscopic endonasal skull base surgeries.1–3 It comprises a 10-year experience including a wide variety of diagnoses and endonasal surgical approaches. Interestingly, we did not encounter any vascular injury during our early experience. We believe that this may be due to the progressive evolution in our technique and indications. Stated differently, we did not tackle complex or high-risk cases until we had matured as a team and until we had developed and mastered hemostatic and reconstructive techniques.
 Prevention of Complications
Prevention of Complications
Prevention of complications starts with a clear understanding of the skull base and vascular anatomy from the endoscopic perspective. The skull base is a relay station where all the cranial nerves and vessels are distributed to their corresponding areas. Our modular endoscopic approaches are based on the identification and control of critical neurovascular structures, especially the internal carotid artery (ICA). From the ventral standpoint the ICAs are brackets that define the lateral limits of the coronal plane. They define the surgical field, which narrows as the ICAs move from proximal to distal (Fig. 45.1). From the endoscopic view, the ICA can be divided into five segments marked by distinct anatomical points (Table 45.2): the parapharyngeal or extracranial segment, the petrous or horizontal segment, the paraclival or vertical segment, the parasellar or cavernous segment, and the supraclinoid segment. Anatomical variations, space-occupying lesions, prior surgery, and varying degrees of pneumatization of the sinonasal tract also influence the position and approach to each segment.
Proper training is also critical for the prevention and management of surgical complications.4 We have proposed guidelines to train endoscopic skull base surgeons, regardless of the specialty, who have to progress through sequential and incremental modules (see Chapter 9). Levels of training are defined by the complexity of the involved anatomy, the technical difficulty associated with the surgery, the potential risk of neurovascular complications, the extent of intradural dissection, and the type of pathology. Proficiency in one level has to be acquired before the surgeon advances to the next. We also developed an animal model for training on endoscopic hemostatic and dissection techniques. This chapter presents the key points regarding vascular control and management of vascular injuries.
 Intraoperative Considerations
Intraoperative Considerations
Interdisciplinary cooperation leading to team surgery is one of the most remarkable advances in skull base surgery. A co-surgeon improves visualization of the surgical field and surveillance of its periphery, increases efficiency, assists with problem solving, provides a continuous second opinion, and counterbalances individual biases.
Neurophysiologic monitoring of cortical and brainstem function is routinely performed. Electromyographic monitoring of cranial nerves is also performed according to the position of the lesion and the required dissection. We advocate using computerized tomographic angiography (CTA) for image guidance, as it highlights the vascular anatomy (except for sellar lesions where we advocate magnetic resonance imaging [MRI]).5 Intraoperative computed tomography (CT) can be used to provide updated information for image guidance and to detect vascular complications such as brain hemorrhage.
Proper surgical technique avoids complications, especially vascular injuries. We strongly advocate bimanual dissection and minimal traction on intracranial tumors to avoid inadvertent avulsion of the surrounding vessels. Following microsurgical principles, the tumor is debulked in a centrifugal direction to allow extracapsular dissection of adjacent neurovascular structures. Hemostasis is achieved using a combination of strategies that include clipping or cauterization of feeding vessels, use of a diamond drill bit for bone removal, application of hemostatic materials, and warm water irrigation.6 Bipolar electrocautery should be used for any intracranial cauterization and whenever a critical nerve or vessel is adjacent to the bleeding site.
Catastrophic bleeding is better prevented than treated. Its potential increases exponentially in the presence of prior surgery, prior radiation therapy, and tumor encasement. Every team should have a plan to follow in case of such occurrence. It is difficult to formulate and execute a plan of action in the middle of a crisis. Significant bleeding from the cavernous sinus and basilar plexus should be anticipated and its management effected by direct application of hemostatic materials and gentle pressure. Staging of a procedure is occasionally warranted for lesions requiring a transclival approach in which the surgeon encounters significant bleeding from the basilar plexus. During the first stage the bone is removed to expose the dura of the posterior fossa, which is then cauterized (bipolar electrocautery) and the basilar plexus is thrombosed with any of the commercially available paste-like hemostatic agents. At this point the surgery can be aborted to stabilize the patient and a second stage is performed 1 to 2 days later.
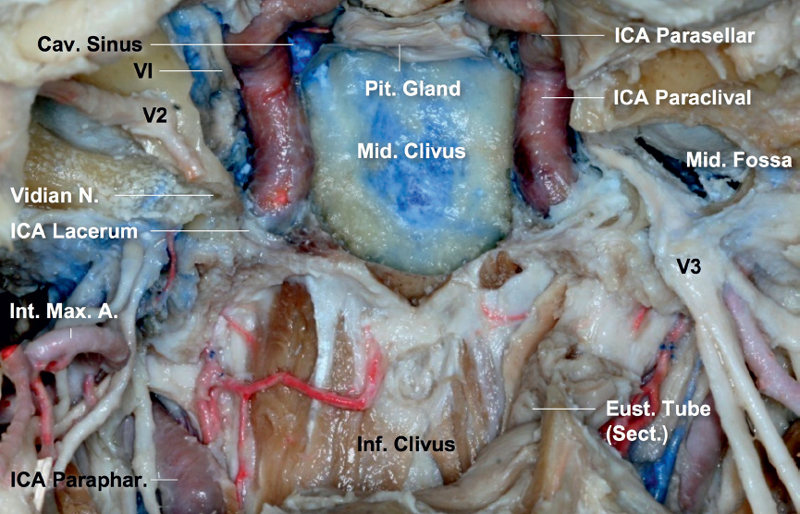
Fig. 45.1 Internal carotid artery (ICA) classification. The ICA can be divided into five segments marked by distinct anatomical points: the parapharyngeal or extracranial segment, the petrous or horizontal segment, the paraclival or vertical segment, the parasellar or cavernous segment, and the supraclinoid segment. The transitional segment between the paraclival and petrosal segments is also called the lacerum segment, as it occupies the upper portion of the foramen lacerum. For communication purposes we consider that the endonasal approaches to expose the area between the ICAs belong to the sagittal plane, and the approaches around the ICA define the coronal plane modules. The latter are anatomically and technically more demanding and carry an increased risk of vascular injury. A.: artery; Cav.: cavernous; Eust.: eustachian; Inf.: inferior; Int.: internal; Max.: maxillary; Mid.: middle; N.: nerve; Paraphar.: parapharyngeal segment; Pit.: pituitary; Sect.: sectioned.




