Fig. 32.1
Anatomy of sacroiliac joint
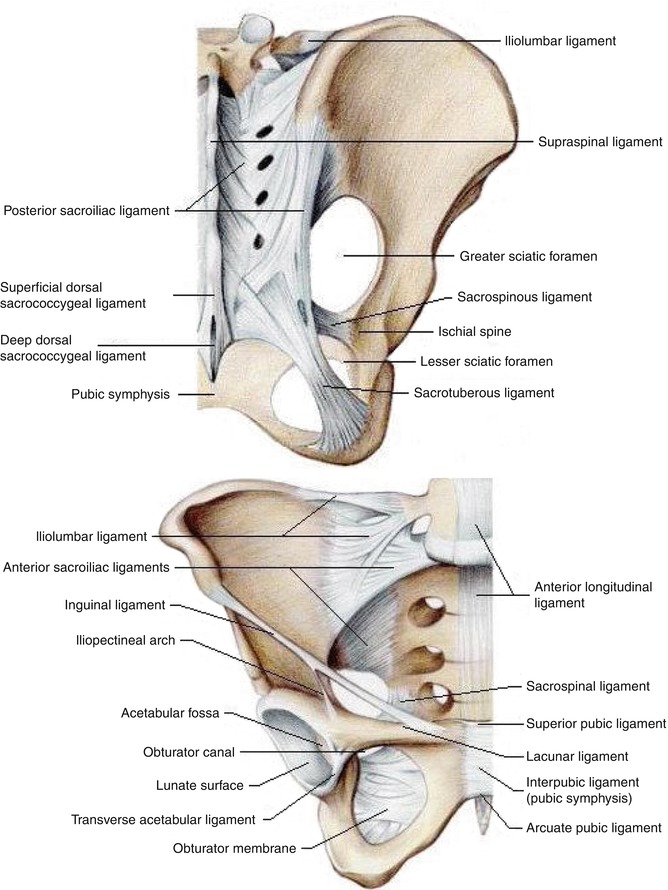
Fig. 32.2
Ligaments of sacroiliac joint
Gender, age, and weight significantly influence the probability that a sacroiliac joint origin contributes to the patient’s chronic low back pain [7]. Females are much more likely to suffer from sacroiliac joint pain than males likely due to the difference in anatomy as well as hormonal and structural changes related to pregnancy and childbirth. Older age correlates with an increased incidence of sacroiliac joint disease likely from the accumulative slow degeneration of the joint with aging. Interestingly, lower BMIs are associated with higher probability of pain from sacroiliac joints if a patient presents with low back pain. This may be simply due to the lower incidence of other low back pain etiologies in patients with low BMIs. According to a multivariant analysis, for an elderly female who presents with chronic low back pain, the chance of the pain originating from the sacroiliac joint could be well above 50 % [7].
Previous lumbosacral spine surgeries predispose patients to the development of sacroiliac joint disease [15, 27]. When lumbar fusion surgeries are extended to the sacrum, the sacral iliac joints are the adjacent joints to the fusion. Therefore, they experience the increased load and stress similar to other spinal adjacent joints after fusion surgery. The increased load and stress from the fused lumbosacral segments might expedite sacroiliac joint degeneration. It has been reported that the incidence of sacroiliac joint degeneration is higher in patients with fusion down to S1 than in patients with fusion down to L5 [15]. In biomechanical studies, it has been shown that posterior fusion of the lumbar spine leads to increased motion at the sacroiliac joint and increased stress across the sacroiliac articular surfaces [6, 16, 19].
A history of iliac crest bone graft harvesting also predisposes patients to sacroiliac joint degeneration. Harvesting cancellous bone was shown to induce pelvic instability [5]. In some studies, history of iliac bone graft harvesting correlated with increased sacroiliac joint degeneration [9].
The probability of chronic low back pain originating from sacroiliac joint degeneration is greater in patients diagnosed with failed back syndrome. Failed back syndrome is characterized by the occurrence or persistence of severe low back pain after spine surgery. A portion of patients with failed back syndrome may have had a misdiagnosis from the start with the sacroiliac joints being the source of pain instead of the lower lumbar spine. Some other patients may have developed new sacroiliac joint degeneration due to the increased stress from the lumbar fusion surgery. As a result, for patients who present with either persistent or new onset low back pain after lumbar surgeries, physicians must have a high suspicion whether the sacroiliac joints are the source of the low back pain.
One of the difficulties of treating sacroiliac joint disease is the lack of accurate diagnostic tests. Numerous sacroiliac joint provocative tests have been described to suggest the pain source, yet the specificity of those tests is generally low [8, 25]. Radiographic tests have been shown to have low sensitivity and specificity in diagnosing sacroiliac joint disease [10]. The most reliable diagnostic test for sacroiliac joint disease is the temporary pain relief after low-volume local anesthetic agents injection into the joint under fluoroscopic control [12, 13, 18, 23] (Figs. 32.3a, b and 32.4). Sacroiliac joint block is considered the gold standard for diagnosing sacroiliac joint disease. Yet, even the sacroiliac joint injection has been shown to be not very reliable. The effects of two consecutive injections are identical only 60 % of the time. One possible explanation for the inaccuracy of the sacroiliac joint injection is the diffusion of the anesthetic agents out of the sacroiliac joint during injection. The diffusion causes the anesthetic agents to come in contact with adjacent nerve trunks or roots, which leads to temporary pain relief even if the sacroiliac joint may not be the sources of pain. Injection of the anesthetic agents into the sacroiliac joint might relieve pain from numerous surrounding ligaments as well.
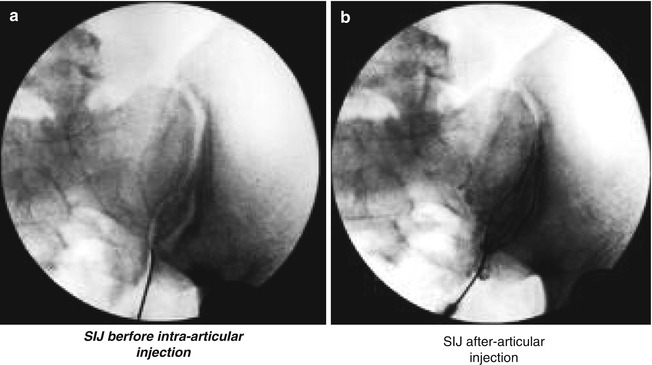
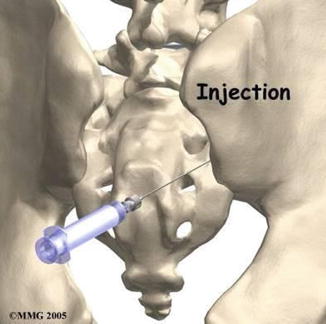

Fig. 32.3
(a) SIJ before intra-articular injection. (b) SIJ after intra-articular injection

Fig. 32.4
Illustration of SIJ injection
Given the uncertainty of diagnosing sacroiliac joint disease, it is without surprise that it is not easy to identify an effective treatment for the disease. The first-line treatments for sacroiliac joint pain are conservative treatments including analgesic and anti-inflammatory medications, physical therapy, and several types of injection treatments. When conservative therapies fail to relieve the symptoms and the physicians believe the pain is originating from the sacroiliac joint, more invasive treatment modalities are considered. Two such treatment options are surgical fusion of the sacroiliac joints or ablative therapy to denervate the joints. A systemic review of the published results from six fusion studies and five denervation ablative studies reported that the majority of patients were satisfied after receiving either treatment [2]. Both procedures reported efficacy in improving pain and functional outcome. However, the evidence was low to very low since all the studies were case series and the number of patients in those studies was generally low. In addition, the effects were relatively moderate. The mean rate of patient satisfaction was 57.6 % for fusion studies with great variations (range from 18 % to 100 %). The mean pain improvement in the studies that reported visual analog or numeric rating showed the improvement in the pain scale of 3.5 and 4.9 points, respectively. One study that documented Oswestry Disability Index (ODI) showed an improvement of 14.0 points. These two treatments (fusion and denervation ablation surgery) have comparable improvements in pain relief and functional outcome. The denervation ablation studies generally have a short follow-up of 6–12 months; therefore, it is unclear whether the denervation procedures provide durable pain relief. It is worth noting that many lumbar facet joint denervation ablation studies showed loss of efficacy after about 2 years.
When conservative treatment fails to relieve pain from suspected sacroiliac joint disease, fusion surgery becomes a treatment option. Various open fusion surgery techniques have been developed including the posterior or Smith-Petersen approach, the anterior approach, and the posterior midline fascia splitting approach [4, 14]. The results from the open sacroiliac joint fusion surgeries were mixed. Some reported great efficacy, yet some results were disappointing. In one study by Schutz et al., the nonunion rate with instability was 41.2 %, and more than 80 % of the patients still had significant pain after the surgery [21]. The open surgery itself is usually painful due to the extensive dissection needed for the surgery. The relatively high complications and nonunion rates of open surgeries encourage surgeons to search for alternatives with possible lower morbidities and higher efficacy.
In recent years, several minimally invasive sacroiliac joint arthrodesis techniques have been developed [1, 17, 20, 26]. Not all the techniques have clinical data to back up their effectiveness yet. However, the limited publications up to date indicated generally good clinical outcomes using minimally invasive sacroiliac joint fusion techniques in treating sacroiliac joint pain that had failed conservative treatments.
In 2008, Wise and Dall described a minimally invasive sacroiliac joint fusion procedure and published their series of 13 consecutive patients [26]. All the patients had no relief after more than 6 months of conservative therapy and completed exhaustive workups to rule out lumbar spine as the source of pain. A fluoroscopically guided intra-articular injection with a mixture of local anesthetic and corticosteroid was used to confirm the diagnosis of sacroiliac joint dysfunction. Their surgical technique involved inserting cages along the anterior-posterior (AP) axis of the sacroiliac joint percutaneously. The key for safe placement of the cages in the AP axis was the understanding of the safe cephalad and caudad margins of the sacroiliac joints and the depth of placement based on preoperative images. It is important to avoid placing the cage too deep through the anterior portion of the joint and into the pelvis. Preoperative CT scans of the bilateral sacroiliac joints with the patients in the prone position (same position as in the surgery) were used to determine the area with the most bony surface available on both sacral and iliac sides of the joint as the “safe zone” for cage placement. During surgery, with the patient in prone position, the starting point of the incision was made on the most prominent part of the posterior superior iliac spine (PSIS) and extended 1 cm caudad and 4 cm cephalad. The dissection was carried down to the PSIS, and a calibrated Steinman pin was tapped into the bone carefully. The placement of the Steinman pin was approximately 6–7 mm cephalad to the caudal margin of the safe zone. The depth of the Steinman pin advancement was carefully monitored using fluoroscopy so that it was within the preoperative measurements (usually between 4.5 and 6.5 cm). When the pin was at the predetermined position, a 9-mm cannulated drill was used to drill over the pin. The surgeon watched the lateral fluoroscopic images carefully to make sure the tip of the pin was not advancing with the drill to avoid its advancement into the pelvis. After palpation of the drilled hole, a threaded titanium cage, measuring 11 × 25 mm (Medtronic Sofamor Danek, Memphis, TN), was then packed with BMP and inserted in the hole with the open slots of the cage’s sidewalls pointing to the sacrum and the ileum. After the first cage was placed, the second cage was inserted in a similar fashion, approximately 6–7 mm cephalad to the first cage. In their series of 13 consecutive patients, an 89 % fusion rate was achieved based on the 6-month CT scan. The low back pain improved by an average of 4.9 points on a ten-point visual analog scale. Leg pain improved by an average of 2.4 points, and dyspareunia improved by an average of 2.6 points. Seventy-seven percent of the patients reported satisfaction with the results of the surgery.
Al-khayer et al. from the UK published their method of percutaneous sacroiliac joint fusion and analyzed their outcomes from nine patients in the same year [1]. In their procedure, the authors used one single hollow modular anchorage (HMA) screw (Aesculap, Sheffield, UK), which is a hollow cylindrical implant made from one piece of titanium alloy and placed the screw perpendicular to and across the sacroiliac joint from the lateral side. The screw passed through the sacral alar to the body of S1 vertebra at the midpoint between the S1 neural foramina and the L5/S1 disc. Patients were placed supine on a radiolucent table. Four views (lateral, anteroposterior, inlet, and outlet) of the pelvis were obtained. A guidewire was placed percutaneously and advanced across the sacroiliac joint to the body of the S1 vertebra under fluoroscopic guidance. A cannulated 10-mm drill was then used to drill over the guidewire with the protection of a guide tube, also under fluoroscopic guidance. The guidewire was then removed, and a 10-mm tap was used to prepare for screw insertion. The HMA screw was then packed with the bone graft from the bone reaming obtained during drilling with demineralized bone matrix and screwed into the prepared tunnel. The usual length of the screw was about 40–50 mm. The position of the screw was confirmed using the fluoroscope. In their series of nine patients with an average follow-up of 40 months, the mean VAS dropped from 8.1 preoperatively to 4.6 postoperatively. The mean ODI dropped from 59 to 45. The mean patient satisfaction was 6.8, and all the patients stated that they would undergo the same procedure again under the same circumstances. Placing the anchoring screws across the sacroiliac joint may have the benefits of immediate stabilization, before the fusion happens. Although the posteriorly placed grafts into the AP axis of the SI joints like the ones used by Wise and Dall may have the same effect functioning as a door wedge stabilizing a pivoting door.
Khurana et al. also reported their experience with 15 patients using the same hollow modular anchorage screws filled with demineralized bone matrix to treat the refractive sacroiliac joint disease [17]. In a mean follow-up of 17 months, the average Short Form-36 scores improved from 37 to 80 for physical function and from 53 to 86 for general health. Thirteen patients (87 %) had good to excellent outcomes.
There are several commercial minimally invasive sacroiliac joint fusion systems available on the market currently. Most of the systems were developed within the past several years; therefore, only limited data is available regarding the clinical efficacy of these systems.
Get Clinical Tree app for offline access

1.
iFuse®: The iFuse Implant System® from SI-BONE® places several (usually three) porous plasma-coated titanium implants across the sacroiliac joints laterally through an incision of approximately 3 cm (Fig. 32.5). The surgical technique includes initial placement of guidewire pins across the sacroiliac joint under fluoroscopic guidance. A drill and a triangular broach are then used to prepare the bone over the pins with the help of a soft tissue protector. The triangular implant is then inserted into the prepared tunnel with a mallet. Usually three implants are placed, in the sacral alar, above or adjacent to the S1 foramen, and between the S1 and S2 foramen, respectively (Figs. 32.6 and 32.7). There are several unique features of the iFuse system. The triangular implant profile minimizes rotations of the implant. The porous surface is designed to minimize micromotion of the implants and promotes bony overgrowth leading to eventual fusion. Usually three implants are placed in one sacroiliac joint; therefore, rotation movements of the sacroiliac joints with the single-threaded cage system should be minimized, and strong immediate stabilization of sacroiliac joint is achieved. According to the company website, biomechanical studies demonstrated that the 7-mm triangular implant is three times stronger in shear and bending strength than an 8-mm cannulated screw. In the iFuse system, no autologous graft, allograft, or other fusion extenders were placed in the sacroiliac joints, nor does the sacroiliac joint be decorticated in preparation for fusion. The eventual bony fusion was therefore dependent upon the porous surface of the implants. Rudolf reported the use of iFuse for minimally invasive sacroiliac joint fusion on the first consecutive 50 patients treated [20]. Pain scale decreased from 7.6 to 3.3 at the 12-month follow-up and to 2.0 at the 24-month follow-up. Eighty-two percent of the patients reported satisfaction of the surgery. No radiographic outcomes about whether the implants led to fusion were discussed. Eleven patients (22 %) experienced complications including three cases of superficial cellulitis, one deep infection, two large buttock hematomas, one nondisplaced ilium fracture, and one delayed loosening of the implants causing recurrence of symptoms. Three patients were taken back to the OR for implant penetration into the sacral neural foramen or L5 neural foramen.










