Chapter 34 Neuropsychology
Goals of Neuropsychology
When neural damage is present or cognitive changes are observed or reported during clinical evaluation, an extended neuropsychological evaluation is appropriate. The prominent neuropsychologist, Arthur Benton (1975), best described neuropsychology as “a refinement of clinical neurological observation [that] serves the function of enhancing clinical observation [and] is closely allied to clinical neurological evaluation and in fact can be considered to be a special form of it.” (p. 68) In clinical settings, neuropsychological assessment aims to extend the clinical neurological exam by: (1) providing important diagnostic information and predictions, even in conditions not detected via other procedures (e.g., anoxia), (2) attributing cognitive strengths and weaknesses to their appropriate factors (e.g., psychiatric symptoms, neurological disease, demographic factors), (3) predicting functional ability to enhance treatment planning, and (4) monitoring cognitive changes and treatment effectiveness across time (Lezak et al., 2004). Neuropsychological assessment is also used in forensic settings and for research in neuroscience, but discussion on these topics is beyond the clinical focus of this chapter.
Before the advent of neuroimaging in the 1970s and 1980s, one of the main goals of neuropsychology was lesion localization. Since current structural imaging techniques are capable of localizing lesions with remarkable accuracy, the focus of neuropsychology has shifted toward characterizing patients’ cognitive and behavioral profiles. Such profiles can be used to make differential diagnosis decisions, especially when lesions may not be evident. For example, some types of dementias present with a cortical profile (i.e., impairments of memory and language), whereas others present with a characteristic subcortical profile (i.e., impairments of processing speed, executive functioning, and mood) (Table 34.1). Repeated neuropsychological evaluations can also be a sensitive method for monitoring the progression of neurodegenerative diseases over time (e.g., Huntington disease [HD], Alzheimer disease [AD]), recovery after an acute injury (e.g., stroke or traumatic brain injury [TBI]), or postoperative recovery following neurosurgery (e.g., temporal lobectomy). Evaluating the effectiveness of medical procedures also entails repeated assessment of cognitive abilities with pre- and post-treatment testing.
Table 34.1 Neuropsychological Characteristics of Cortical versus Subcortical Dementia Using Alzheimer Disease and Huntington Disease as Examples
| Alzheimer Disease (Cortical Dementia) | Huntington Disease (Subcortical Dementia) | |
|---|---|---|
| LEARNING AND MEMORY | ||
| Episodic memory | Impaired encoding/consolidation | Impaired information retrieval |
| Poor delayed recall and recognition memory | Recognition memory is better than delayed recall | |
| Retrograde amnesia | Severe, temporally graded, retrograde amnesia | Mild, non-graded, retrograde amnesia |
| Priming | Impaired | Preserved |
| Implicit procedural/motor learning | Preserved | Impaired |
| Implicit cognitive skill learning | Preserved | Impaired |
| ATTENTION/CONCENTRATION | Relatively preserved | Poor auditory and visual attention |
| PROCESSING SPEED | Relatively intact | Very slow |
| EXECUTIVE FUNCTIONING | ||
| Set shifting | Better able to shift focus | Difficulty with perseveration |
| Working memory | Mild deficits in ability to manipulate information, but preserved phonological loop and visuospatial sketchpad | Early notable deficits in phonological loop, visuospatial sketchpad, and ability to manipulate information |
| LANGUAGE AND SEMANTIC KNOWLEDGE | ||
| Speech | Preserved | Dysarthric and slow |
| Fluency | More impaired semantic fluency than phonemic fluency | Severe and equal impairment in phonemic and semantic fluency |
| Naming | Impaired; more semantic errors (e.g., calling a lion “an animal”) | Relatively preserved; more perceptual errors (e.g., calling a bucket “a cup”) |
| Structure of semantic knowledge | Tend to focus on concrete perceptual information | Able to focus on abstract conceptual knowledge |
Adapted from Salmon, D.P., Filoteo, J.V., 2007. Neuropsychology of cortical versus subcortical dementia syndromes. Semin Neurol 27, 7-21.
Neuropsychological Evaluation
Test Administration
Two major approaches to neuropsychological evaluation currently dominate the field: the fixed battery approach and the flexible battery approach (Barr, 2008). The fixed battery approach requires that the same tests are administered to every patient in a standardized manner. One example of a fixed battery is the Halstead-Reitan battery (Box 34.1), for which comprehensive norms have been published by Heaton and colleagues (Heaton et al., 1991). An advantage to the fixed battery approach is that the information gathered is comprehensive and systematically assesses multiple domains of cognitive functioning. Additionally, if repeated assessments are available, test scores can be directly compared with baseline information, and tests are well validated and normed. Drawbacks of the fixed battery approach include its length (up to 8 hours), because it may be too long for some patients to tolerate and is difficult to afford with the limited reimbursement schedules in managed care. Furthermore, an extended assessment may not be necessary to address the referral question.
Box 34.1
Heaton Adaptation of Halstead-Reitan Neuropsychological Test Battery
Adapted from Heaton, R.K., Grant, I., Matthews, C.G., 1991. Comprehensive Norms for Expanded Halstead-Reitan Battery: Demographic Corrections, Research Findings, and Clinical Applications. Psychological Assessment Resources, Odessa, Florida.
In contrast to the fixed battery approach, the flexible battery (or hypothesis-driven) approach allows neuropsychologists to develop a test battery based on the referral question, the patient’s history, and the clinical interview. In the flexible battery approach, a brief set of basic tests is initially administered, and additional tests of more specific abilities are used to conduct in-depth follow-up assessments based on each particular patient’s needs. For example, clinicians using the Iowa-Benton method (Tranel, 2008) specifically tailor testing to each patient based on their presenting concern by administering the appropriate portions of a core battery, which are then followed up with tests that assess suspected impairments in more detail (Fig. 34.1). Considerations when selecting tests include age, primary language, level of education, ethnicity/cultural factors, reading level, expected level of global cognitive impairment (to avoid ceiling or floor effects in testing), and physical disabilities (Smith et al., 2008). Although this approach is more tailored to the individual needs of the patient (and is therefore briefer), it can be less comprehensive than the fixed battery approach. Most neuropsychologists’ approaches fall somewhere between the use of a set battery and a completely individualized examination.
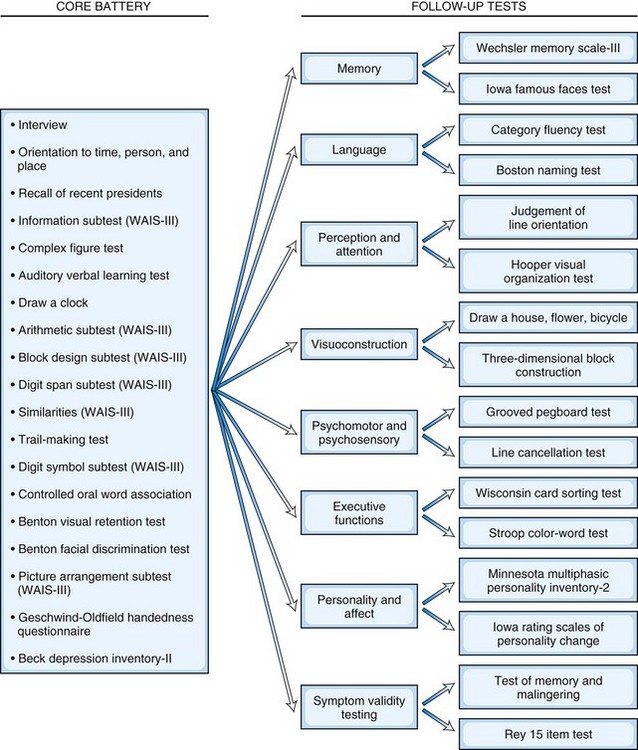
Fig. 34.1 Example of a flexible battery approach.
(Adapted from Tranel, D., 2008. Theories of clinical neuropsychology and brain-behavior relationships, in: Morgan, J.E., Ricker, J.H. (Eds), Textbook of Clinical Neuropsychology. Taylor & Francis, New York, pp. 25-37.)
Test Interpretation
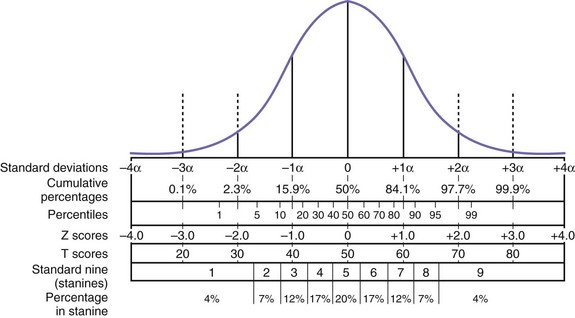
Neuropsychological test scores are interpreted most often by using group statistics to make inferences about individual patients through the use of normative data, which are typically collected by test developers as a standardization sample. Normative data aid in test interpretation by accounting for variables that are likely to influence test performance (e.g., demographic factors) so that accurate and appropriate conclusions are drawn. Confounding variables are accounted for by stratifying test scores according to gender, age, and/or level of education. An individual’s raw score is compared with the distribution of scores from his or her peer group to determine where it falls within the range of expected performances. Fig. 34.2 and Table 34.2 show a normal distribution and interpretive guidelines for use in neuropsychological interpretation. The usefulness of normative data depends strongly on the size and representativeness of the standardization sample. Clinical interpretation can also be greatly affected by the goodness-of- fit between the individual patient and the standardization sample. For example, it would not be appropriate to make determinations about the test performance of an 82-year-old man with 8 years of education by comparing his test score with those of a group of 40-year-olds with an average of 12 years of education. Furthermore, it is important to use the most recent norms available, because cohort effects may lead to differences between current patients and those from whom data were collected years ago. When appropriate norms are not available, there is a danger of overdiagnosis or underdiagnosis of cognitive impairment. Accurate interpretation of neuropsychological test performance necessarily incorporates information about the sample from which the test norms were developed.
Table 34.2 Descriptive Terms Associated with Performance within Various Ranges of the Normal Distribution
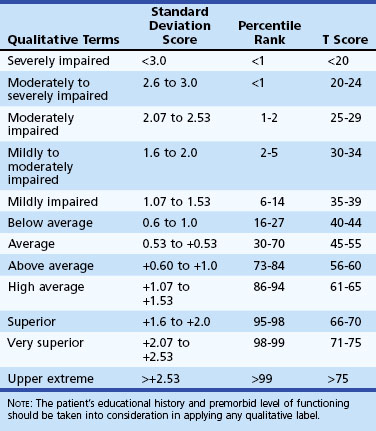
Another approach to test interpretation is through the use of cut scores. Tests that rely on cut scores often measure performances with low base rates or deficits very few healthy people demonstrate. Some tests are fairly straightforward in their capability to measure abilities that are largely intact in normal subjects but impaired in disordered patients. For example, most people are able to bisect a line without difficulty, but patients with left-sided visuospatial neglect typically identify the midpoint of the line to be to the right of center. Other tests, however, are more complex and require more sophisticated analyses to develop valid cut scores. Smith et al. (2008) provide an excellent explanation for how cut scores are useful individual statistics (i.e., test scores) that allow inferences about which diagnostic group a patient is likely to belong to (e.g., AD versus mild cognitive impairment versus healthy). Test validation studies commonly use sensitivity and specificity data and base rate information to calculate likelihood ratios and positive predictive values for individual tests. Likelihood ratios and positive predictive values are differing expressions of the probability that a patient has a particular condition given his or her test score. Smith et al. (2008) put these concepts another way by saying, “the positive predictive value allows for statements such as: ‘Based on the patient having earned a score of y on test z, the probability that this patient has the condition of interest is x.’ ” (p. 47). A common example of the application of cut scores can be found in the use of screening instruments to quickly identify potential impairments. For example, a cut score of 23 on the Mini-Mental State Examination (MMSE) has frequently been used as an indicator of cognitive impairment for dementia. Although cut scores can save time and provide useful heuristics, these scores may miss “true hits” and identify “false positives.”
The comparison of current performance with past test scores is another important component of test interpretation, especially if cognitive decline is suspected. Rarely, however, do individuals have previous test data available for these comparisons. When no previous test scores are available, evidence of the patient’s premorbid intellectual functioning is estimated. Several techniques are available for estimating premorbid intellect. Measures known as hold tests (i.e., cognitive tests that are resistant to neurological insult), such as reading ability measures, are frequently used because they are more resistant to many processes that cause decline in cognitive functioning (Smith-Seemiller et al., 1997). Some common measures of reading ability are the National Adult Reading Test (Nelson, 1982), Wide Range Achievement Test Revision 4 (Wilkinson and Robertson, 2006), and Wechsler Test of Adult Reading (Psychological Corporation, 2001), all of which assess the ability to read irregularly spelled words aloud. In some cases, reading ability measures may not be appropriate estimates, because they are affected by the progression of disease (e.g., dementia) (Cockburn et al., 2000; Taylor, 1999). In such conditions, estimates that use demographic variables (e.g., age, education level, primary occupation) in regression-based formulas to predict premorbid IQ may be more useful (e.g., the Barona formula; see Box 34.2). Similarly, academic records (e.g., college grade point average, achievement tests from middle school) can provide estimates of functioning prior to illness or injury. Most contemporary neuropsychologists use a combination of these strategies, either formally (e.g., Oklahoma Premorbid Intelligence Estimate-3) (Schoenberg et al., 2002) or informally, to arrive at the best estimate of premorbid ability.
Box 34.2
Barona Regression-Based Premorbid IQ Estimation Formula
Barona premorbid IQ estimation formula: VIQ = 54.23 + 0.49 (age) + 1.92 (sex) + 4.24 (race) + 5.25 (education) + 1.89 (occupation) + 1.24 (region)
Age: 16 to 17 years = 1; 18 to 19 years = 2; 20 to 24 years = 3; 25 to 34 years = 4; 35 to 44 years = 5; 45 to 54 years = 6; 55 to 64 years = 7; 65 to 69 years = 8; and 70 to 74 years = 9
Race: black = 1; other ethnicity = 2; white = 3
Education in years: 0 to 7 years of school = 1; 8 years = 2; 9 to 11 years = 3; 12 years = 4; 13 to 15 years = 5; and 16 or more years of school = 6
Occupation: farm laborers, farm foremen, and laborers (unskilled workers) = 1; operatives, service workers, farmers, and farm managers (semiskilled) = 2; not in the labor force = 3; craftsmen and foremen (skilled workers) = 4; managers, officials, proprietors, clerical, and sales workers = 5; professional and technical = 6
Region: Southern region = 1; North Central region = 2; Western region = 3; Northeast region = 4
Adapted from Barona, A., Reynolds, C., Chastain, R., 1984. A demographically based index of premorbid intelligence for the WAIS-R. J Clin Consult Psychol 52, 885-887.
Brief Mental Status Examination
Mini-Mental State Examination
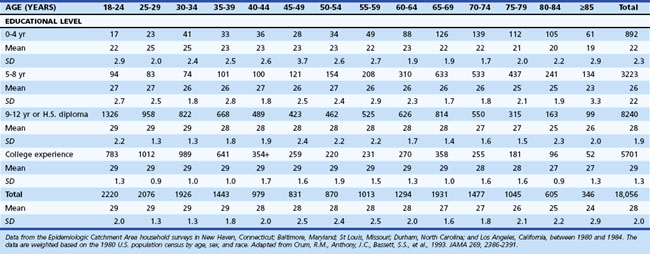
One of the most widely used mental status examinations is the MMSE (Folstein et al., 1975), a 30-point standardized screening tool for assessing orientation, attention, short-term recall, naming, repetition, simple verbal and written commands, writing, and construction (Fig. 34.3). The MMSE has been used in a variety of settings (e.g., community, institutions, general hospital, specialty clinics), with many different neurological and psychiatric conditions (e.g., dementia, stroke, depression), across age ranges, and with different cultural and ethnic subgroups. Demographic variables such as age and education have been shown to systematically influence MMSE scores, so normative data or cut scores should account for these variables. One example of appropriate norms comes from the Epidemiologic Catchment Area study (Crum et al., 1993); these are presented in Table 34.3. Whereas many intact individuals achieve total scores near 30, a cut score of 23 on the MMSE has been shown to have adequate sensitivity and specificity (86% and 91%, respectively) for detecting dementia in community samples (Cullen et al., 2005). However, when working with highly educated patients (i.e., ≥16 years of formal education) a cut score of 27 is recommended. A cut score of 27 has correct classification rate of 90.1% and a likelihood ratio of 9.6, which means that highly educated individuals with cognitive complaints and MMSE scores below 27 are almost 10 times more likely to have dementia than those with scores greater than 27 (O’Bryant et al., 2008). MMSE scores can also track changes across time. Longitudinal studies have found patients with AD show an average annual rate of change of 2.81 points, although change rates are not uniform across illness stages or gender (Chatfield et al., 2007).

Fig. 34.3 Mini-Mental State Examination.
(Reprinted with permission from Folstein, M.F., Folstein, S.E., McHugh, P.R., 1975. Mini-Mental State: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12, 189-198.)
Table 34.3 Mini-Mental State Examination Score by Age and Educational Level, Number of Participants, Mean, Standard Deviation, and Selected Percentiles
Despite its widespread use, the MMSE has some drawbacks. One potential threat to the test’s internal validity is the non-standardized administration of some of the items. Examples of these frequent adaptations of the MMSE include the use of nonorthogonal (i.e., semantically related) word stimuli for registration and recall, nonstandard scoring of serial 7s, and nonstandard inclusion of spelling world backwards. Standard scoring instructions can alleviate some of these problems. Another drawback of the MMSE is that it has “ceiling effects” that can miss cognitive impairments in high-functioning individuals. The MMSE also has difficulty differentiating individuals with mild cognitive impairment (MCI) from controls and those with dementia (Mitchell, 2009). Finally, because this test relies on a single total score, partial administration of the measure (e.g., due to sensory impairments of the patient) provides no information about cognitive status.
Modified Mini-Mental State Examination
Some of the criticisms of the MMSE led to the development of the Modified Mini-Mental State Examination (3MS) (Teng and Chui, 1987), a 15-item extension of the MMSE that assesses orientation (self, time, place), attention (simple and complex), memory (recall and recognition), language (naming, verbal fluency, repetition, following commands, writing), construction, and executive functioning (similarities). It remains relatively brief to administer (10 minutes), and age- and education-corrected normative data are available (Tschanz et al., 2002). Regression-based prediction formulas for the 3MS allow for more accurate assessments of change across time (Tombaugh, 2005). The broader scoring range (0 to 100) has been shown to be more sensitive than that of the MMSE in identifying dementia (McDowell et al., 1997; Tschanz et al., 2002) and other cognitive disorders (Bland and Newman, 2001) in large community samples. A cut score for cognitive impairment is typically 77 (Bland and Newman, 2001; McDowell et al., 1997), and a change of 5 points over the course of 5 to 10 years indicates the presence of clinically meaningful decline (Andrew and Rockwood, 2008).
Montreal Cognitive Assessment
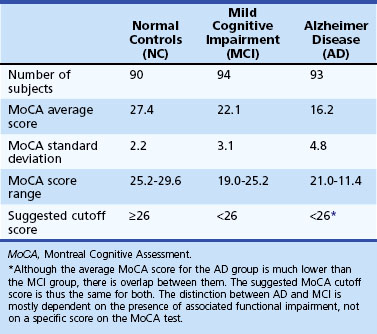
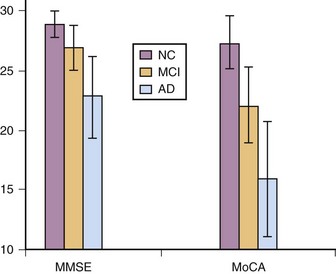
The Montreal Cognitive Assessment (MoCA) was originally developed as a screening tool to correct the MMSE’s insensitivity to MCI (Nasreddine et al., 2005). Executive functioning, immediate and delayed memory, visuospatial abilities, attention, working memory, language, and orientation to time and place are all assessed in this one-page measure (Fig. 34.4). The total score ranges from 0 to 30 points, and a cut score of 26 has demonstrated very good specificity (by correctly identifying 87% of healthy participants) and excellent sensitivity when differentiating MCI (90%) and AD (100%) from healthy comparisons (Table 34.4). More importantly, the positive predictive value of the MoCA was 89% for both MCI and AD. The psychometric properties of the MoCA contrast with the MMSE’s sensitivity of 18% for MCI and 78% for AD (although the MMSE had a specificity of 100% in the same study, meaning it correctly ruled out dementia in all cases) (Fig. 34.5). Studies in Parkinson disease (PD) (Hoops et al., 2009; Nazem et al., 2009) and HD (Videnovic et al., 2010) populations have also shown that the MoCA has promise as a measure sensitive to early stages of different types of dementia. The MoCA has the advantage of assessing more cognitive domains, therefore reducing the likelihood that impairments or disorders would be overlooked (e.g., executive dysfunction, a hallmark symptoms of vascular dementia). Another advantage is that the test is free to clinicians (http://www.mocatest.org) and has been translated into 31 different languages and dialects.

Fig. 34.4 Montreal Cognitive Assessment.
(Reprinted with permission from Nasreddine, Z.S., Phillips, N.A., Bedirian, V., et al., 2005. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53, 695-699.)
Telephone Interview for Cognitive Status—Modified
The Telephone Interview for Cognitive Status—Modified (mTICS) is a relatively brief screening instrument that was designed to quickly and accurately assess cognition over the telephone, although it can also be used in face-to-face settings. This 13-item measure is heavily weighted toward immediate and delayed free recall, which might make it particularly useful in detecting mild impairments (Duff et al., 2009; Lines et al., 2003) such as amnestic MCI (Fig. 34.6). Age-, education-, gender-, and race-corrected normative data are available (Hogervorst et al., 2004).


Fig. 34.6 Modified Telephone Interview for Cognitive Status.
(Data from Welsh, K.A., Breitner, J.C.S., Magruder-Habib, K.M., 1993. Detection of dementia in the elderly using telephone screening of cognitive status. Neuropsychiatry Neuropsychol Behav Neurol 6, 103-110.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree