Fig. 24.1
Schematic drawing of the lumbar plexus from the lateral view (Courtesy of Dr. Juan S. Uribe)
A number of cadaveric studies have detailed the anatomy of the lumbar plexus in relation to the psoas major muscle and the lumbar vertebral bodies. These studies have generally divided the vertebral bodies into “safe working zones,” defined by the absence of a crossing lumbar plexus branch. Four zones comprise the vertebral bodies: Zone 1 is the most ventral quarter of the vertebral body, Zone II is the middle anterior quarter, Zone III is the middle posterior quarter, and Zone IV is the most dorsal quarter (Fig. 24.2). One of the first of these cadaveric studies was conducted by Benglis et al. [3] in 2009 using three cadavers. These authors found that the lumbosacral plexus lies on the dorsal surface of the psoas muscle, within a cleft formed between the junction of the transverse process and vertebral body. The plexus was most dorsally positioned at L1–L2 (at the posterior endplate, i.e., Zone IV, at this level) and was found to migrate ventrally then caudally down the lumbar spine. The safe zones identified by this study were Zones I–III at L1–L2, L2–L3, and L3–L4 and Zones I–II at L4–L5. The authors thus suggested positioning the dilators and tubular retractors at the anterior half of the disc spaces.
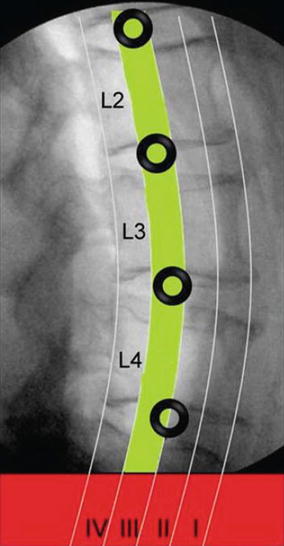

Fig. 24.2
Lateral radiograph of the lumbar spine demonstrating division of the vertebral bodies into four zones (Zones I–IV) from anterior to posterior. The relative “safe zone” (Zone III) is depicted in green. The recommended safe working zones to prevent direct nerve injury are indicated with black circles at each level (Courtesy of Dr. Juan S. Uribe)
In a more comprehensive analysis of 20 cadavers, Uribe et al. [4] found that all parts of the lumbar plexus, including the nerve roots, were contained within the substance of the psoas muscle dorsal to Zone IV. An important exception to this was the genitofemoral nerve, which originates at L1–L2, courses obliquely along Zone II at L2–L3, emerges superficially and anterior from the medial border of the psoas muscle at L3–L4, and courses along Zone I at L4–L5. Thus, the authors refined the safe zones of Benglis et al. to be Zone III at L1–L2, L2–L3, and L3–L4 and the midpoint of the vertebral body (Zone II/III demarcation) at L4–L5.
Two other cadaveric studies have further modified the safe working zones at each lumbar disc space. In a study of eight cadavers, Guerin et al. [5] found the safe zones to be Zones II–III at L1–L2, Zone III at L2–L3 and L3–L4, and Zone II at L4–L5. An early study of 30 cadavers by Moro et al. [6] found the safe zones to be Zones I–III at L1–L2 and L2–L3; these authors were concerned about injury to the genitofemoral nerve anywhere near the L3–L4 and L4–L5 disc spaces.
Analyzing the aforementioned data, the strictest safe working zones are as follows:
It deserves noting that Guerin et al. expressed significant concern regarding the sheer quantity of neurovascular structures at L4–L5 and advocated that other routes of access (transforaminal lumbar interbody, posterior lumbar interbody, or anterior lumbar interbody) be considered at that level.
In an important study, Dakwar et al. [7] dissected six cadavers in order to detail the anatomy of the lumbar plexus branches outside of the psoas muscle, i.e., in the retroperitoneum and abdominal wall musculature. The authors identified four nerves particularly at risk during retroperitoneal dissection: the subcostal, iliohypogastric, ilioinguinal, and lateral femoral cutaneous nerves. These nerves are also at risk for retraction injury. In order to minimize the risk of injury, the authors advocated “sequential and gentle muscle dilation with blunt instruments (hemostat forceps) until the vertebral column is identified.” They also emphasized “early identification of the posterior wall of the retroperitoneum (quadratus lumborum muscle) and gentle dissection of the space from posterior to anterior and superior to inferior until the transverse process and the psoas muscle are identified at the target level, to avoid injury to the main nerves that run freely in the retroperitoneal cavity.”
Injury to the nerves that supply the abdominal wall can result in abdominal flank bulge; thus, understanding this relevant anatomy is also essential. The abdominal wall consists of four major muscle groups: the rectus abdominis, transverse abdominis, and external and internal oblique muscles. The T11 and T12 intercostal nerves provide most of the innervation to the abdominal wall musculature [8], specifically via the subcostal nerve, which travels underneath the transversus abdominis muscle to innervate the rectus abdominis and external oblique muscles (Fig. 24.3) [7]. The iliohypogastric and ilioinguinal nerves also provide innervation, specifically to the internal oblique and transverse abdominis muscles. Special care must be taken to avoid severing, cauterizing, suturing, or otherwise injuring these nerves during exposure and closure.
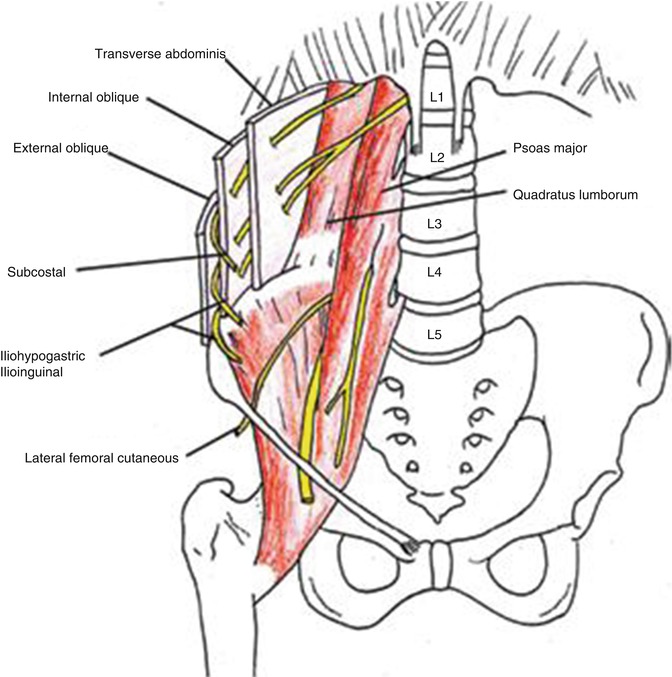

Fig. 24.3
Illustration demonstrating the trajectory of the four main nerves traveling outside the psoas muscle in the retroperitoneum along the posterior abdominal wall and within the abdominal muscles
Anatomical positioning for the minimally invasive lateral retroperitoneal transpsoas approach is worth mentioning (Fig. 24.4). Patients are placed in the lateral decubitus position with right-side down, and an axillary roll protects the axilla. The dependent (right) arm is supinated and outstretched on an arm rest with the elbow padded. The nondependent (left) arm is pronated and raised, also with the elbow padded. In a series of 1,000 consecutive spine surgeries with patients in five different positions, Kamel et al. [9] found that the lateral decubitus position was associated with the highest incidence (7.5 %) of position-related upper extremity somatosensory-evoked potential (SSEP) changes. This was thought attributable to vertical forces that compress the brachial plexus and the first rib. Thus, adequate padding and keeping the patient’s limbs in unstressed positions is paramount to minimizing iatrogenic nerve injury.
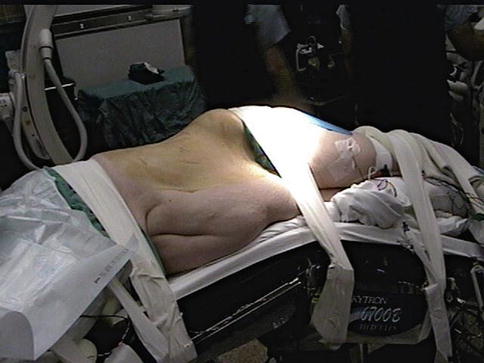

Fig. 24.4
Patient in the lateral decubitus position with the table broken
24.3 Types of Monitoring
The three standard intraoperative neuromonitoring modalities used during spinal surgery are SSEPs, motor-evoked potentials (MEPs), and electromyography (EMG). Each of these modalities has been proposed to play an important role at specific stages of surgery. During spinal surgery, it has been suggested that neurophysiological testing helps to avoid unforeseen neuronal or vascular injury, reduces the risk of a permanent postoperative deficit, and provides localization of specific nerve roots. SSEPs comprise signals recorded from multiple positions along the afferent pathway of primarily proprioceptive tracts. These are transmitted via the dorsal columns of the spinal cord. As mentioned above, when patients are positioned laterally, SSEPs can be particularly useful in possibly identifying and reversing impending damage to peripheral nerves within both upper and lower extremities (by repositioning the extremity of concern) [9]. A peripheral disturbance in neuronal transmission may be resultant of stretch or compression in any of the extremities in this lateral position. As an example, the peripheral nerve(s) in the top arm, primarily at the site of the brachial plexus, can be stretched if proper padding techniques are not administered. The bottom arm can also experience similar strain as nerve(s) can be compressed if the head is not positioned properly in a neutral fashion and/or an axillary roll is inappropriately placed. Furthermore, compression of the ulnar nerve can occur in either of the arms if they are not adequately padded. The break in the surgical table can cause the bottom leg to be compressed or the top leg to be stretched past its functional threshold. It is of utmost importance for the neuromonitoring team to have reliable peripheral SSEP responses (recorded from Erb’s point in the upper extremities and the popliteal fossa in the lower extremities) to distinguish between changes caused by positioning, surgical manipulation, pharmacological events, or underlying physiological issues.
MEPs are elicited by transcranial activation of the motor cortex and are transmitted through the lateral corticospinal tracts of the cord, exiting at each nerve root level, continuing transmission of the signal to the motor axons that innervate each of the designated muscles. MEPs have been proposed to be a very sensitive test for assessment of motor function and may allow for more rapid identification of potential damage to neural structures. Although further study is needed, some practitioners feel that effective MEP methods and techniques can give additional prognostic information for the assessment of patient outcomes.
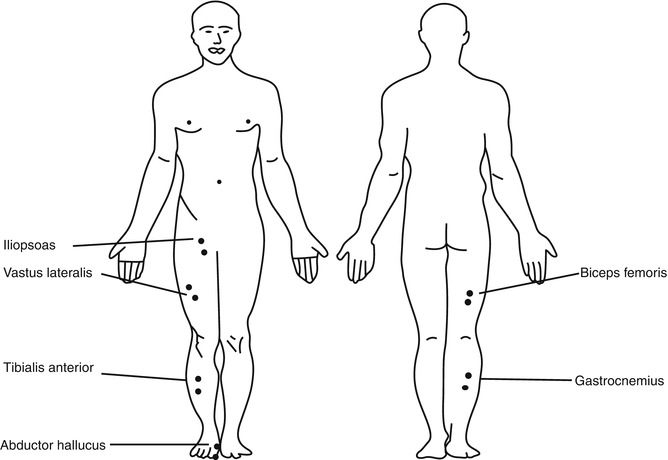
From a technical perspective, EMG electrodes are placed in the muscles that supply the nerve(s) that may be at risk during a given procedure. For the lateral approach, these muscles include the iliopsoas, vastus lateralis, biceps femoris, tibialis anterior, gastrocnemius, and abductor hallucis muscles (Fig. 24.5). Depending upon the neuromonitoring group’s protocol, the electrodes used to monitor MEP responses may be sufficient to also monitor the EMG. However, in general, more extensive coverage of muscles is applied to EMG monitoring than is to MEP monitoring. It should be noted that any electrodes used to monitor EMG can theoretically be used to record MEP responses as well; two additional cortical stimulating electrodes are placed to add MEPs as a modality when requested. Both SSEP and EMG monitoring are limited in the information each can give the surgical and neuromonitoring teams (such as an ischemic event at an individual root level). Thus, MEPs may provide the surgeon with more data that can assist with surgical decision-making.