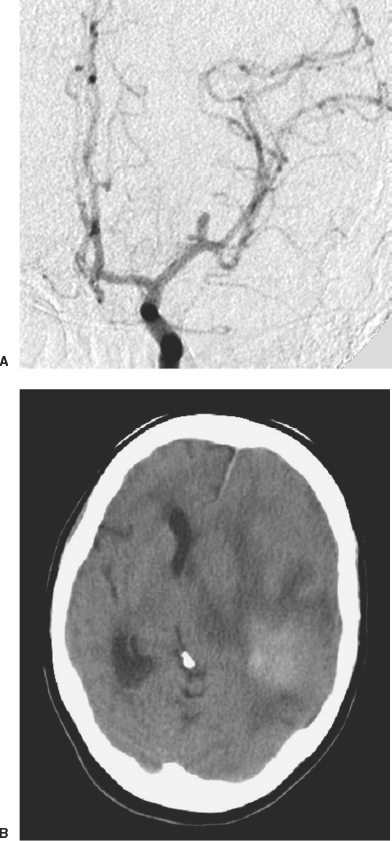
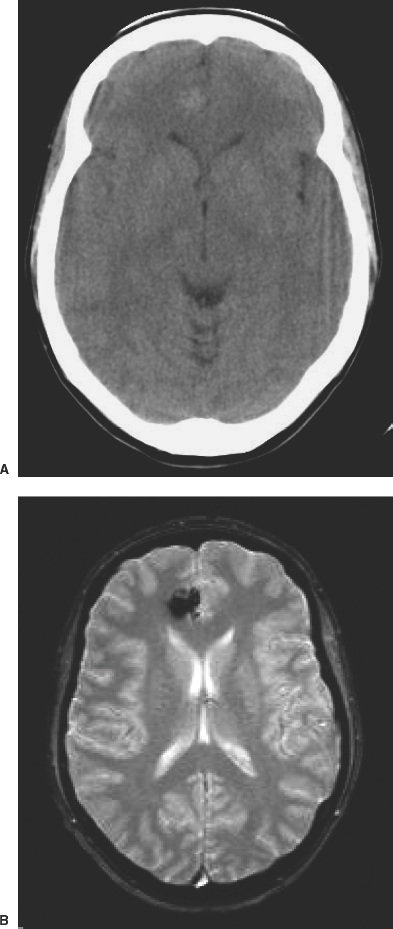
7 Spontaneous Intracerebral Hemorrhage Patrick Mitchell, Barbara A. Gregson, Raghu R. Vindlacheruvu, and A. David Mendelow It has long been known from postmortem studies that some strokes are caused by intracerebral hemorrhage (ICH), but a reliable premortem diagnosis was rarely made in the days before the introduction of computed tomography (CT) scanning in 1975.1 Since the early 1980s CT and magnetic resonance (MR) scanning have become widely available so that now almost everybody presenting with a stroke has one of these investigations. This has made it possible to study the natural history of ICH and to try specific medical and surgical treatments. The treatment that has received most attention is surgical evacuation of the hematoma, but despite 30 years of research and 12 completed randomized trials, it remains uncertain whether surgical clot evacuation brings any benefits. The largest trial completed to date was the Surgical Trial in ICH (STICH), the result of which was published in 2005.2 The STICH Trial spanned a period from 1993 to 2004 and recruited 1033 patients who were randomized to receive “initial conservative management” or “early surgery.” Its neutral result has been instrumental in dimming enthusiasm for surgical evacuation of hematomas. When this result is meta-analyzed with those of the other completed randomized trials, the overall conclusion remains neutral. These results notwithstanding, it would be an oversimplification to dismiss surgery as a treatment for ICH. Several hypotheses of benefit from surgery have survived the results of these trials. Specifically, in some cases, the mechanistic argument for removing a clot is strong and such cases were not well represented in trials because of a lack of equipoise in the minds of the treating surgeons. Superficial lobar hematomas, not complicated by extension into more central areas or the ventricles, are one such group. There is an ongoing research initiative around the world to address some of these hypotheses, with answers expected over the next 5 years. The most useful classification scheme for ICH is based on etiology, as this is directly related to treatment options. Intracranial hemorrhages fall into two broad groups: those that arise from “ictohemorrhagic” vascular lesions and those that do not. The lesions concerned are those that can be diagnosed with currently available imaging techniques. There are a wide range of ictohemorrhagic lesions that can cause ICH: they all have in common the risk that they may bleed again in the future. This means that treating an ICH that has arisen from an ictohemorrhagic source involves the two aims of dealing with the acute hemorrhage and dealing with the cause to prevent any further hemorrhages in the future. The lesions involved are arteriovenous malformations (AVMs), cavernous malformations (CVMs), aneurysms, dural fistulae, and tumors. Most spontaneous ICHs that present do not arise from an underlying macroscopic ictohemorrhagic lesion. They arise either from microaneurysms in the brain parenchyma, known as Charcot Bouchard aneurysms that are associated with hypertension, or from amyloid angiopathy that is a common component of a range of neurodegenerative disorders. In parallel with this etiological classification, an anatomical classification is also useful. The deficit inflicted by a hemorrhage is closely dependent on the eloquence of the brain area in which it arises. The most serious disabilities arise from hemorrhages in the left hemisphere, particularly those that are deep-seated. An important anatomical distinction is made between hemorrhages in the supratentorial compartment and those in the posterior fossa. The posterior fossa is much smaller than the supratentorial compartment and contains tightly packed eloquent vital centers. Most posterior fossa clots occur in the relatively noneloquent cerebellum and exert their clinical effects by compression rather than direct destruction of the brainstem. They also have a marked tendency to produce hydrocephalus. These factors lead to strong mechanistic arguments for surgical evacuation of posterior fossa clots in patients who deteriorate, and they have generally not been included in trials of ICH. Arteriovenous malformations are congenital vascular abnormalities that involve shunting of blood between arteries and veins within the brain. They are present from birth and can give rise to hemorrhages at any age. Overall most intracranial hemorrhages occur later in life and so those presenting in younger people are generally more likely to arise from AVMs. Other clinical features can give clues that a presenting ICH is of AVM origin. They are prone to provoke epilepsy and steal phenomena which may be associated with a history of neurological deficit or focal epileptic activity in a similar distribution to the presenting deficit. Because AVMs are low-pressure lesions located in brain tissue that is already abnormal, when they hemorrhage they tend to give less deficit than one would expect from the size of the clot (Fig. 7-1). There are certain systemic conditions associated with cerebral AVMs that carry both cutaneous or ocular features such as the Sturge-Weber or von Hippel-Lindau syndromes. Even in the absence of a specific syndrome, evidence of cutaneous vascular malformations on the head is a suggestive clue to an underlying AVM. Many AVMs are associated with calcification that can be seen on CT scans and this is a strong clue to an underlying AVM, if seen with an ICH. Equally sometimes serpiginous enhancing areas are seen that correspond to large draining veins. Definitive diagnosis is made with catheter angiography or an alternative angiographic modality such as MR or CT. Untreated AVMs carry an annual risk of hemorrhage that ranges from 2% to 4%.3,6 These hemorrhages are associated with a morbidity of between 38% to 53% and a mortality of 10% to 18%,7–10 significantly lower than the rates for other ICHs. When treatment of an AVM is undertaken these risks are not eliminated or even significantly reduced unless the AVM is completely removed or obliterated and cannot be seen on catheter angiography.11–14 It has been reported that the hemorrhage rate may actually increase following treatment if complete elimination is not achieved.14 Three modalities are available to treat AVMs: surgical excision, stereotactic radiosurgery, and endovascular embolization. There are some large or complex AVMs that cannot be completely obliterated even with a combination of all three of these treatments, and in such cases it is better not to embark on treatment at all. This means that it is necessary to consider whether a particular AVM has a reasonable chance of being cured before starting treatment, and the need for this judgment as well as the number of modalities involved means that large or complex AVMs are best treated in a multidisciplinary setting. Figure 7-1. Left frontal spontaneous intracerebral hemorrhage with associated subdural hemorrhage seen on a CT scan (A). This arose from an AVM that was shown on a catheter angiogram (B). The hemorrhage was managed conservatively. Elective microsurgical resection of the AVM was done after 3 weeks when the hematoma had liquified. AVM, arteriovenous malformations. Surgical excision has the potential to eliminate the hemorrhage risk immediately. It has the problem of a significant operative morbidity and mortality, especially for larger and more complex lesions or those located in eloquent areas.7,18 A particular problem with surgery is the so-called Normal Perfusion Pressure Breakthrough Phenomenon.8,9 The arteriovenous shunting effect of AVMs lowers the cerebral perfusion pressure in their immediate vicinity. This longstanding lowering of perfusion pressure leads to dilated and fragile vasculature around the AVM. If the AVM is surgically removed, local perfusion pressure is restored to normal and the fragility of this vasculature leads to a significant rate of postoperative swelling and hemorrhage formation. Like surgery, endovascular embolization has the potential to completely obliterate an AVM and afford immediate protection from hemorrhage. It is also considerably safer than surgery, with lower associated morbidity and mortality. Its main problem is that as a single treatment it has a low obliteration rate of 0% to 22%.21–23 This low obliteration rate makes it a poor standalone treatment, but in a multidisciplinary setting it is frequently used as a primary treatment because it is quite often possible to tailor embolization to enhance the success rate of other treatments rather than to achieve a cure. Cases where cure is achieved can be seen as a bonus. For example, it is often possible specifically to embolize deep arterial feeders, which are particularly difficult to control surgically.22 Or, alternatively, it may be possibly to embolize more diffuse peripheral areas of an AVM leaving a compact nidus as a suitable target for stereotactic radiosurgery.24 We are still at a relatively early stage in the evolution of embolization technology, with new products and techniques becoming readily available. This holds the promise of improvements in the efficacy of the technique in the future. Focused radiosurgery involves administering a single dose of radiotherapy that is accurately focused on the AVM. There are two groups of technology available for doing this. Linear accelerators use a single narrow X-ray beam that passes through the target but is moved through an arc so outwith the target the radiation exposure is spread over wider areas of brain. The Gamma Knife® is a single product produced by Elekta. It focuses 201 beams of gamma rays from cobalt sources onto a small target. Of these technologies the Gamma Knife is considered to have the longest track record and best characterized clinical efficacy in treating AVMs. The treatment involves attaching a stereotactic frame to the patient’s head, usually under local anesthetic. The patient is then imaged with the frame in place and the imaging data used to relate the geometry of the AVM to the frame. The frame is then used accurately to target the radiation dose to the lesion. Stereotactic radiosurgery obliterates between 65% and 85% of AVMs that are less than 3 cm in diameter.13,25,26 With larger AVMs the total radiation dose to the surrounding brain per Gray delivered to the target is increased and it is necessary to reduce the target dose accordingly. This leads to a reduced obliteration rate in larger lesions.27–29 The technique is best suited to AVMs with small compact nidi rather than more diffuse ones. A significant limitation of stereotactic radiosurgery is that it takes between 1 and 4 years following the treatment for the AVM to be obliterated, so a hemorrhage risk persists for this time period. Figure 7-2 Aneurysmal hematoma associated with subarachnoid hemorrhage, arising due to rupture of a right middle cerebral artery aneurysm. Most cerebral aneurysms lie outside the brain parenchyma in the subarachnoid space. When they rupture they usually give rise to a subarachnoid hemorrhage, which is a clinical entity that is distinct from ICH. Frequently a subarachnoid hemorrhage will be complicated by extension of blood into the parenchyma, but in such cases management is usually dictated by the subarachnoid hemorrhage overall rather than the intraparenchymal component. Less frequently an aneurysm will give rise to an ICH with little or no subarachnoid hemorrhage. Such ICHs are often devastating because aneurysms are high-pressure, high-flow lesions that are located proximally. A history of sudden severe headache, followed by collapse, and imaging findings of a hemorrhage that is adjacent to one of the proximal cerebral vessels are clues that there may be an underlying aneurysm (Fig. 7-2). Aneurysmal ICHs are unusual in that there is good evidence for clinical benefit for prompt surgical evacuation.17,18 If an operation to remove the hematoma is being undertaken, the aneurysm can be isolated from the circulation to prevent future hemorrhage by applying a spring-loaded clip across its neck. This method of securing aneurysms has been available for the past 50 years, but since the early 1990s an alternative treatment of endovascular embolization with fine, coiled platinum wire has been developed. In the context of subarachnoid hemorrhage coiling is the better tolerated of the two treatments but it is not quite so effective at preventing rehemorrhage.19 For this reason it is now favored in the treatment of aneurysms that have caused a subarachnoid hemorrhage, but in the context of ICH, when an operation is being done anyway, the argument is much weaker. Nevertheless, in the UK there is a trend toward coiling aneurysms at the time when they are demonstrated on angiography so that there is no need for dissection around the cerebral arteries, with associated morbidity (Fig. 7-3).33 Figure 7-3 This ruptured left middle cerebral artery aneurysm was treated by coiling. Remarkably the hematoma was well tolerated by the patient. However, there was an acute deterioration 11 days later, comprising right-sided hemiplegia and aphasia, with impairment of conscious level. The hematoma was evacuated without need for the aneurysm to be dissected out. The patient responded well to this treatment. Cavernous malformations are small nodular vascular lesions with a relatively low blood flow that cannot be seen on intraarterial angiography. They are indistinct at best on CT but are easily seen on MRI because they contain paramagnetic hemoglobin breakdown products of various ages (Fig. 7-4). They occur throughout the central nervous system and carry a variable risk of hemorrhage, ranging from some that cause repeated clinically significant hemorrhages to some that remain asymptomatic indefinitely. The hematomas they cause tend to be small and morbidity is dependent on the eloquence of the area in which the CVM is located.34,35 Devastating hemorrhages are rare and tend to occur with CVMs in the optic chiasm, midbrain, brainstem, or spinal cord. Because the hemorrhages are small, surgical evacuation in the acute phase is rarely justified, but once a lesion has bled it tends to denote a high risk of recurrent hemorrhage in the future and surgical evacuation to prevent such recurrences should be considered, especially if the lesion has bled more than once.34,36,37 The true nature of unruptured asymptomatic CVMs is unknown. Because they cannot be seen on angiography, embolization is not a treatment option, but stereotactic radiosurgery has been used. It seems to reduce the hemorrhage rate by about three quarters rather than give total protection from rebleeds and, as with AVMs, there is a time lag of more than 1 year before the fall in rebleed rate is seen. There have been some reported cases of multiple CVMs38 and of their sometimes being associated with previous radiotherapy.39 There may also be an association with venous malformations which themselves do not bleed at all. Dural fistulae are shunts between arteries and veins like AVMs, but unlike AVMs they are located in the dura and associated with the dural venous sinuses rather than in the brain. Their origin is unclear but in some cases they appear to arise secondary to trauma. They may be provoked by venous sinus thrombosis and are probably not congenital. They are associated with localized raised venous pressure and a tendency to intracerebral and subdural hemorrhage. Treatment modalities available are as for AVMs: endovascular embolization, surgery, and stereotactic radio surgery,40 though in the case of stereotactic radiosurgery, their peripheral location may make them inaccessible to this modality if they occur in somebody with a large head at the frontal or occipital poles.
Classification of Intracerebral Hemorrhage
Arteriovenous Malformations
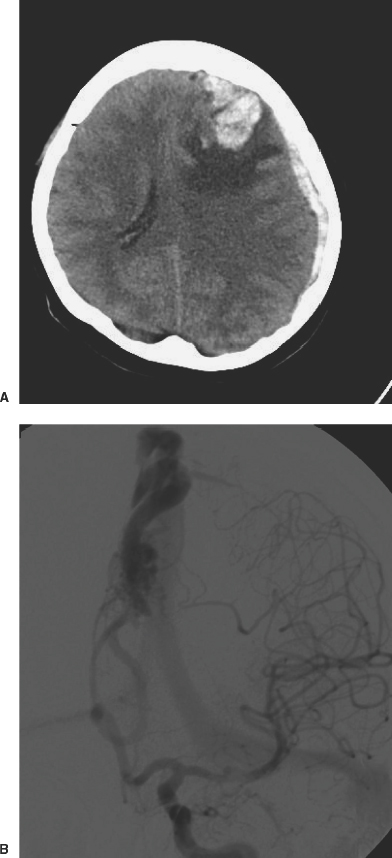
Surgical Excision
Embolization
Focused Radiosurgery
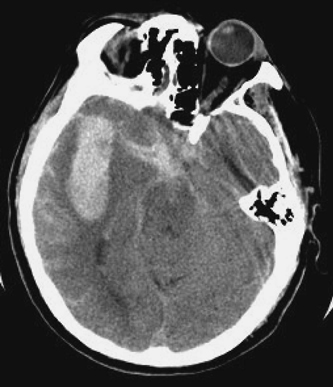
Aneurysms
Cavernous Malformations
Dural Fistulae
Spontaneous Intracerebral Hemorrhage
Only gold members can continue reading. Log In or Register to continue

Full access? Get Clinical Tree








