INTRODUCTION
Subarachnoid hemorrhage (SAH) accounts for 5% of all strokes; it affects nearly 30,000 people each year in the United States, with an annual incidence of 1 per 10,000. Saccular (or berry) aneurysms at the base of the brain cause 80% of all cases of SAH. Nonaneurysmal causes of SAH are listed in
Table 39.1. SAH most frequently occurs between ages 40 and 60 years, and women are affected more often than men.
SAH due to the rupture of an intracranial aneurysm is a devastating event; approximately 12% of patients die before receiving medical attention, and another 20% die after admission to the hospital. Of the two-thirds of patients who survive, approximately one-half remain permanently disabled, primarily due to neurocognitive deficits and depression. Advances in neurosurgery and intensive care, including an emphasis on early aneurysm clipping and aggressive therapy for vasospasm, have led to improved survival over the last three decades, with a reduction in overall case fatality from approximately 50% to 20% in high-volume centers.
PATHOLOGY AND EPIDEMIOLOGY OF INTRACRANIAL ANEURYSMS
Saccular aneurysms most often occur at the circle of Willis or its major branches, especially at bifurcations. They arise where the arterial elastic lamina and tunica media are defective and tend to enlarge with age. The typical aneurysm wall is composed only of intima and adventitia and can become paper thin. Many aneurysms, particularly those that rupture, are irregular and multilobulated, and larger aneurysms may be partially or completely filled with an organized clot, which occasionally is calcified. The point of rupture is usually through the dome of the aneurysm.
Eighty-five percent to 90% of intracranial aneurysms are located in the anterior circulation, with the three most common sites being the anterior communicating artery complex (approximately 40%), the junction of the posterior communicating and internal carotid artery (approximately 30%), and the middle cerebral artery at the first major branch point in the sylvian fissure (approximately 20%). Posterior circulation aneurysms most often occur at the apex of the basilar artery or at the junction of the vertebral and posteroinferior cerebellar artery. Saccular aneurysms of the distal cerebral arterial tree are rare. Nearly 20% of patients have two or more aneurysms; many of these are “mirror” aneurysms on the same vessel but on the contralateral side.
Intracranial aneurysms are uncommon in children but occur with a frequency of 2% in adults, suggesting that approximately 2 to 3 million Americans have an aneurysm. However, more than 90% of these are small (< 10 mm) and remain asymptomatic throughout life. The annual risk of rupture of an asymptomatic intracranial aneurysm is approximately 0.7%. Important risk factors for the initial rupture of an aneurysm include increasing size, prior SAH from a separate aneurysm, and basilar apex and posterior communicating artery location (
Table 39.2). The most powerful of these risk factors is size: for example, internal carotid artery aneurysms < 7 mm in diameter bleed at a rate of approximately 0.1% per year, compared to an annual rate of 8% for those >25 mm in size. Other risk factors for aneurysm rupture in approximate order of importance include cigarette smoking, aneurysm-related headache or cranial nerve compression, heavy alcohol use, a family history of SAH, female sex (especially when postmenopausal), multiple aneurysms, hypertension, and exposure to cocaine or other sympathomimetic agents. In deciding whether to treat an unruptured intracranial aneurysm, the risks associated with repair should always be balanced against the patient’s estimated lifetime risk of hemorrhage without repair.
The prevalence of aneurysms increases with age and is higher in patients with atherosclerosis, a family history of intracranial aneurysm, or autosomal dominant polycystic kidney disease (PCKD). Intracranial aneurysms have also been associated with Ehlers-Danlos syndrome, Marfan syndrome, pseudoxanthoma elasticum, coarctation of the aorta, and sickle cell disease. Screening for unruptured intracranial aneurysms with magnetic resonance angiography (MRA) is indicated in patients with PCKD and in family members who have two or more first-degree relatives with intracranial aneurysms; testing is positive in 5% to 10% of these individuals.
CLINICAL FEATURES
SAH usually commences with an explosive “thunderclap” headache followed by stiff neck. The pain is often described as “the worst headache of my life.” The headache is usually generalized, but focal pain may refer to the site of aneurysmal rupture (e.g., periorbital pain related to an ophthalmic artery aneurysm). Commonly
associated symptoms include loss of consciousness, nausea and vomiting, back or leg pain, and photophobia. In patients who lose consciousness, tonic posturing may occur and may be difficult to differentiate from a seizure. Although aneurysmal rupture often occurs during periods of exercise or physical stress, SAH can occur at any time, including sleep.
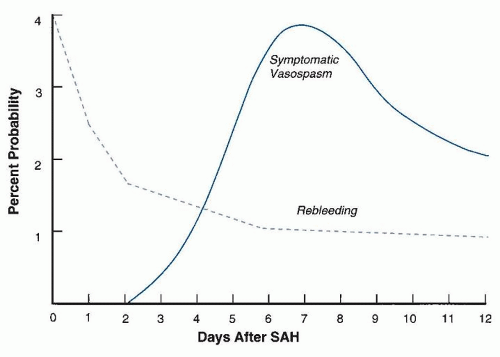
More than one-third of patients give a history of suspicious symptoms days or weeks earlier, including headache, stiff neck, nausea and vomiting, syncope, or disturbed vision. These prodromal symptoms are often due to minor leaking of blood from the aneurysm and are therefore referred to as “warning leaks” or “sentinel headaches.” Initial misdiagnosis of SAH occurs in approximately 15% of patients, and those with the mildest symptoms are at greatest risk. Approximately 40% of misdiagnosed patients experience subsequent neurologic deterioration due to rebleeding, hydrocephalus, or vasospasm before reaching medical attention, with increased morbidity and mortality.
Stiff neck and the Kernig sign are hallmarks of SAH. However, these signs are not invariably present, and confusion and low back pain are sometimes more prominent than headache. Preretinal or subhyaloid hemorrhages—large, smooth-bordered, and on the retinal surface—occur in up to 25% of patients and are practically pathognomonic of SAH.
The most important determinant of outcome after SAH is the patient’s neurologic condition on arrival at the hospital. Alterations in mental status are the most common abnormality; some patients remain alert and lucid; others are confused, delirious, amnestic, lethargic, stuporous, or comatose. The modified Hunt and Hess grading scale serves as a means for risk stratification for SAH based on the first neurologic examination (
Table 39.3). Patients classified as grade I or II SAH have a relatively good prognosis, grade III carries an intermediate prognosis, and grades IV and V have a poor prognosis. Focal neurologic signs occur in a minority of patients but may point to the site of bleeding; hemiparesis or aphasia suggests a middle cerebral artery aneurysm, and paraparesis or abulia suggests an aneurysm of the proximal anterior cerebral artery. These focal signs are sometimes due to a large hematoma, which may require emergency evacuation.
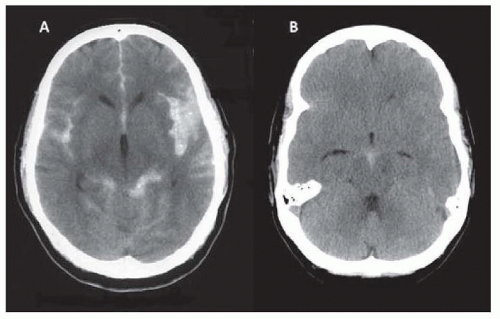
In 10% of patients with nontraumatic SAH and in two-thirds of those with negative angiography, computed tomography (CT) reveals blood confined to the perimesencephalic cisterns, with the center of bleeding adjacent to the midbrain and pons (
Fig. 39.1). Patients with “perimesencephalic” SAH always have normal findings on neurologic examination and a benign clinical course; rebleeding and symptomatic vasospasm or hydrocephalus almost never occur. The source of hemorrhage in these patients is presumably venous.
Symptoms and signs of an unruptured intracranial aneurysm may result from compression of adjacent neural structures or thromboembolism. These aneurysms are often, but not always, large or giant (> 25 mm). Aneurysms of the posterior communicating artery frequently compress the oculomotor nerve (almost always affecting the pupil). Aneurysms of the intracavernous segment of the internal carotid artery may damage the third, fourth, fifth, or sixth cranial nerves, and their rupture can lead to formation of a carotid-cavernous fistula. Less often, large aneurysms compress the cortex or brain stem, causing focal neurologic signs or seizures. Thrombosis within the aneurysmal sac occasionally sends emboli to the artery’s distal territory, causing transient ischemic attacks or infarction. In the absence of SAH, some patients experience sudden, severe headache without nuchal
rigidity, perhaps related to aneurysmal enlargement, thrombosis, or meningeal irritation; these symptoms can clear with aneurysm clipping.