Fig. 2.1
Proliferation of neuronal stem cells and interkinetic nuclear migration
Intermediate progenitor (IP) cells reside within the VZ and often divide at ventricular surface at early stages of neurogenesis (Franco and Muller 2013; Noctor et al. 2004). However, as neurogenesis proceeds, the IP cells migrate to a distinct proliferation layer adjacent to the VZ, the SVZ. Retroviral labeling and time-lapse imaging in embryonic rodent cortical slice cultures as well as staining for neuron markers was used to demonstrate that IP cells most often undergo one round of symmetric division to produce two neurons (Attardo et al. 2008; Haubensak et al. 2004; Kriegstein and Noctor 2004; Noctor et al. 2008). In contrast to RG cells, IP cells seem to lack apical-basal polarity (Attardo et al. 2008; Kriegstein and Noctor 2004; Miyata et al. 2004; Noctor et al. 2004). The ‘two-step pattern’ of neurogenesis, involving RG cells and IP cells, appears to be the predominant principle for cortical neurogenesis in rodents (Haubensak et al. 2004; Kriegstein and Noctor 2004; Miyata et al. 2004). It has been suggested that the emergence of the SVZ and its constituent IP cells may have been responsible for the evolutionary increase in cortical thickness and layering that presumably occurred in the interval between a reptile-like mammalian ancestor and early mammals (Cheung et al. 2007). As mentioned above, neurons are usually born in a position, which differs from their terminal destination. Thus, neurons need to migrate from their place of birth to their final position using several types of cellular mechanisms.
2 The Cellular Mechanisms of Neuronal Migration
The formation of the central nervous system (CNS) depends on two crucial early development events: (1) the proliferation and differentiation of neural stem cells as discussed above, leading to generation of a variety of different types and numbers of neurons; (2) the migration of postmitotic neurons from the VZ and SVZ to appropriate areas and specific locations within the central nervous system where these neurons establish functional neural circuits with each other (Marin and Rubenstein 2003). During central nervous system development, neurons utilize three modes of migration: radial migration, tangential migration, and chain migration (Marin and Rubenstein 2003). In the developing cortex, later-born projection neurons migrate radially along the elongated fiber of radial glia (RG) cells to reach their final destinations at the interface between cortical plate and marginal zone (Hatten 2002; Kriegstein and Noctor 2004; Noctor et al. 2001; Rakic 2007; Tamamaki et al. 2001). Meanwhile, GABAergic cortical interneurons born in the ganglionic eminences migrate tangentially into the developing cortical wall (Anderson et al. 1997; Ang et al. 2003; de Carlos et al. 1996; Marin and Rubenstein 2003). In the postnatal brain, the radial migration mode still remains the primary way for laminar positioning of newly generated granule neurons of the dentate gyrus of the hippocampus (Nowakowski and Rakic 1979). Interestingly, olfactory interneurons migrate from the SVZ of the lateral ventricles in the adult brain to the olfactory bulbs via the rostral migratory stream (RMS), by a specific mode of migration named chain migration (Lois and Alvarez-Buylla 1994; Lois et al. 1996) (see also Chap. 9).
Within the CNS, despite differences in migratory pathways and migration modes among the distinct neuronal subtypes, it is believed that most migratory processes are driven by similar cell-intrinsic mechanisms, and determined by extracellular cues to a large extent. Neurons contain a heterogeneous network of filamentous structures known collectively as the cytoskeleton consisting of the actin microfilaments, the neurofilaments (called intermediate filaments in non-neuronal cells), and the microtubules. Actin is prominent in axons, and is particularly abundant in growing tips of axons, the growth cones. It plays a critical role in orchestrating dynamic changes of cellular morphology. Microtubules form long scaffolds that extend the full length of the neuron, and they also take part in cell movement and cell division. Neurofilaments are the bones of the cytoskeleton and the most abundant fibrillar components of the axon. They are long filaments of approximately 10 nm in diameter, intermediate in size between actin filaments (about 5 nm) and microtubules (about 20 nm). Unlike microtubules, neurofilaments are very stable and remain mostly polymerized within neurons.
Additionally, the extracellular matrix (ECM) acts as a major extracellular signaling mechanism influencing the development of the central nervous system (see also Chap. 9). The ECM is composed of five classes of macromolecules – collagens, elastin, proteoglycans, hyaluronan, and adhesive glycoproteins, such as laminins, reelin, tenascins, etc. The ECM plays important roles during CNS development by acting as a mechanical support, by providing essential survival signals and by regulating neuronal migration, which will be further discussed.
Neuronal migration is a cyclical multi-step process that consists of collectively interrelated but independent discrete events, including four major cell biological stages: polarization, protrusion, adhesion, and retraction (Lauffenburger and Horwitz 1996; Pollard and Borisy 2003; Ridley et al. 2003). Each individual neuron responds to certain extracellular signaling stimuli and the cellular migration process is rapidly initiated. The migration process is coordinated by internal and external signaling mechanisms allowing the cell to form transient specialized structures that permit each neuron to complete the entire cell migration process. Specifically, the neuron first needs to polarize in a certain direction, to form an active and extensive leading edge that allows the dynamic protrusions of neurite to move forward and initiate the cell migration cycle. Subsequently, somal translocation, the most characteristic feature of neuronal migration, involves two consecutive steps. The first step in somal translocation is the formation of a cytoplasmic swelling in the leading edge, immediately ahead of the nucleus. This cytoplasmic dilatation has been observed in both tangentially and radially migrating neurons (Bellion et al. 2005; Konno et al. 2005; Schaar and McConnell 2005). The second step is nucleokinesis, which repositions the nucleus forward into the cytoplasmic dilatation following the centrosome by coordination of many cytoskeletal and signaling molecules (Bellion et al. 2005; Godin et al. 2012; Solecki et al. 2009; Tsai and Gleeson 2005) (Fig. 2.2). The leading edge of migrating neuron displays diverse morphologies in different neuronal types, for instance, the cortical tangentially migrating interneurons dynamically integrate their two leading edge branches into the migratory cycle (Martini et al. 2009; Okada et al. 2007). In contrast, radially migrating neurons seem to have a single leading edge migrating along radial glia fibers (Gupta et al. 2003; Rakic 1972). Parallel to these events, rearrangements in adhesive complexes that link the ECM to the cytoskeleton can lead to attachment and stabilization of neurons to the surrounding substrates, which is very helpful to move forward. Cells modulate adhesion by controlling the surface density, and state of activation of their adhesion receptors. A variety of extracellular stimuli activate intercellular signaling pathways and cytoskeleton components in neurons, which enhance or inhibit the ligand-binding activity of the adhesion receptors to influence cellular adhesion function. As the cell moves forward, the trailing edge must remain in the rear and retract to enable the cell to advance. During neuronal migration, cytoplasmic rearrangements and organelle repositioning also participate in this process. The nucleus is the most remarkable organelle during the forward movement of the soma, and this process is commonly referred to as nucleokinesis (Bellion et al. 2005; Godin et al. 2012; Solecki et al. 2009; Tsai and Gleeson 2005).
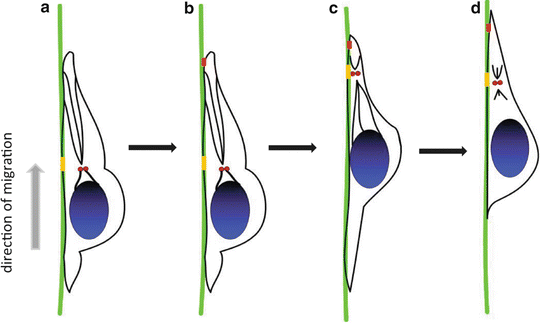

Fig. 2.2
Multi-step process of neuronal migration. (a) Cell movement in a certain direction and formation of the leading edge, (b) Attachment of the leading edge to surrounding substrates, (c) Remodeling of microtubules and nuclear translocation, (d) Retraction of the trailing edge
The cytoskeleton is the major intrinsic determinant of the shape and migration mode of a neuron. Actin filaments play a central role in the leading process formation, intracellular rearrangement events and all of the mechanical steps during the migration cycle, since the actin meshwork can provide the major driving force for cell movement. In addition, other cytoskeletal systems are also required for cell migration. As such, a mechanical role for microtubules is also important during cell movement. For example, microtubules also grow during the elongation of the leading edge, and nuclear movement seems to involve the participation of microtubules (Tanaka et al. 2004). Additionally, microtubules also associate with some important signaling proteins to control neuronal migration. CDK5, a serine/threonine cyclin-dependent kinase, modulates nucleokinesis through phosphorylation of many microtubule-associated proteins, including Lissencephaly (LIS1) and Doublecortin (DCX), which play well-established roles in nucleokinesis (see also Chaps. 1, 5 and 6).
The ECM signals define the timing, the direction, and the final destination for the migrating neurons. The coordination of ECM and cytoskeleton can initiate cell polarization and provide grounds for neuronal migration and lamination by affecting different modes of cellular migration, such as radial, tangential and chain migration, in distinct ways and controlling specific aspects of neuronal migration. For example, laminins is an ECM glycoprotein that have active roles in promoting neuronal migration through binding to cell surface receptors such as integrin and then transducing information to the cytoskeleton (Belvindrah et al. 2007; Chen et al. 2009; Mobley et al. 2009; Stanco et al. 2009). Reelin is an extracellular molecule secreted by Cajal-Retzius cells, and its binding to its receptors induces a series of phosphorylated signaling cascade that regulates microtubule dynamics and triggers neurons to migrate into their proper destination in the cortex (Beffert et al. 2004; D’Arcangelo et al. 1999; Gonzalez-Billault et al. 2005; Hiesberger et al. 1999; Howell et al. 2000). The Reelin signaling pathway is one of the most well-known signaling mechanisms involved in the assembly of the cortical cytoarchitecture (see also Chaps. 1 and 9). Reelin, the product of the Reln gene, is secreted by Cajal-Retzius (CR) cells in the marginal zone (MZ) and binds to the transmembrane lipoprotein receptors APOER2 and VLDL, which are expressed by migrating neurons and RG cells. The promotion of neuronal detachment from RG fibers is a major function of Reelin (Franco et al. 2011). Disruptions in the Reelin signaling pathway in mice cause disorganized lamination while in human causing lissencephaly and cerebellar hypoplasia (Hong et al. 2000). A multistep mechanism has been proposed based on recent research, in which Reelin orients multipolar neurons to polarize their migration by activating RAP1/DAB1, then controlling N-cadherin function to regulate somal translocation (Franco et al. 2011; Jossin and Cooper 2011).
3 The Molecular Mechanisms of Neuronal Migration
Neurons within the developing neocortex find their final destination specifically through a rearrangement of their cytoskeleton in response to extracellular cues mediated by various intracellular signaling pathways (Ayala et al. 2007). In this part, we will discuss the extracellular and intracellular signal mechanisms that regulate the behavior of the cytoskeletal components (Fig. 2.3). Extracellular molecules and surrounding cellular architecture, which function corporately as a microenvironment, play a critical role in neural migration in an inside-out fashion to their respective lamina of neocortex. In the CNS, ECM glycoproteins display a dynamic expression pattern in developing and adult brain (Franco and Muller 2011). The interactions between neuroblasts and ECM molecules are dynamic and mediated via cellular receptors and molecules. In detail, cell surface receptors for ECM are integrins while ECM molecules include Reelin, laminin, proteoglycans and tenascin, which take profound effect in neural migration and lamination in the developing neocortex (Barros et al. 2011). In addition to the glycoproteins mentioned above, the secreted netrins and slits function in axonal outgrowth and guidance, controlling cell adhesion, neuronal migration and polarity (Bradford et al. 2009; Ypsilanti et al. 2010).
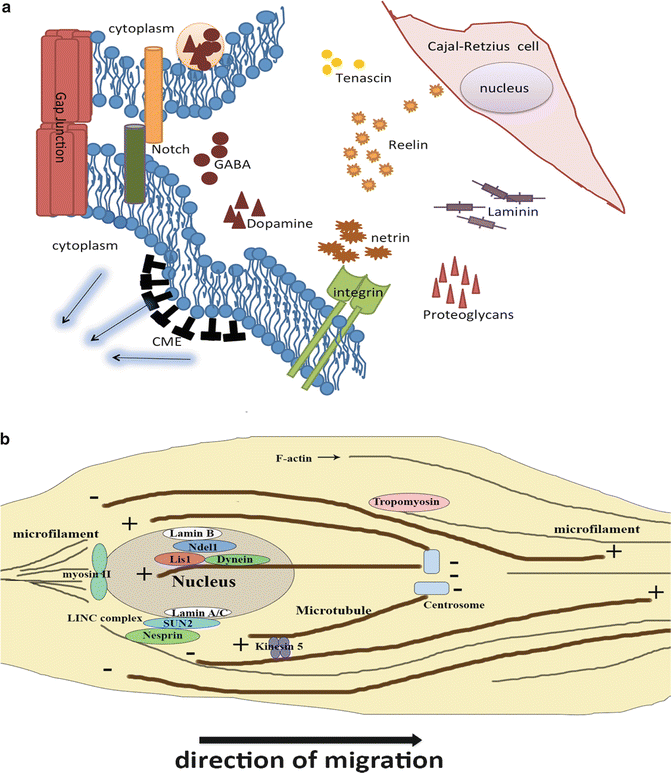

Fig. 2.3
The molecular mechanism of neuronal migration. (a) Molecules in ECM secreted by surrounding cells regulate neuronal migration. Notch signaling pathway and endocytosis are involved in some locomotion regulation. (b) Cytoskeletons, including microfilament and microtubule, and attached molecules play a role in pushing nucleus and cytoplasm to move forward
Cellular communication can influence neuronal migration in the neocortex by regulating neuroblast behavior. Cell communication can occur in a cell-cell contact fashion. It has been suggested that cell-cell communication and/or adhesion through gap-junction-mediated interaction by connexin 43 plays a crucial mechanistic role in radial migration of cortical projection neurons (Elias et al. 2007); and in switching cell migration from tangential to radial manner to allow interneurons to move to their correct laminar position (Elias et al. 2010). Furthermore, cell communication can also capitalize on paracrine signaling in neocortical migration. Chemokines, a family of secreted cytokines, function in neuronal migration during developing brain in addition to their roles in pathological states. To give an example, stromal cell-derived factor 1 (SCD1) reinforces the motility of neuroblasts, migrating from the SVZ towards the olfactory bulbs by upregulating epidermal growth factor receptor (EGFR) and α6 integrin in nearby cells, resulting in enhancement of their ability to bind to laminin in the vascular niche (Asensio and Campbell 1999). Moreover, neurotransmitters are implicated in functioning in modulating the migration of cortical neurons as well (Heng et al. 2007). Dopamine can influence tangential migration of cortical GABAergic neurons by redistributing cytoskeletal elements (Bhide 2009; McCarthy et al. 2007). GABA transiently released near target destinations for migrating neurons, acts as a chemoattractant during corticogenesis by modulating cortical neuronal movement via multiple classes of receptors (Owens and Kriegstein 2002). Blockage of GABAB receptors with a specific antagonist, results in altered tangential migration of cortical interneurons (Lopez-Bendito et al. 2003). TorsinA plays a role in GABAergic neuron migration in the embryonic brain by tethering the nucleus to the cytoskeleton (McCarthy et al. 2012).
The intracellular mechanisms initiated by the extracellular cues are in critical operation during neocortical migration. In addition to the signaling pathways mentioned above, there are additional critical intracellular signal pathways. Notch signaling, which is widely known as a vital regulator of neural stem cells and neural development, affects neuronal migration by altering the morphology of migrating neurons. Increased Notch signaling leads to a bipolar morphology that favors migration, while decreased Notch signaling leads to a more multipolar morphology that stalls migration (Hashimoto-Torii et al. 2008). Notch signal regulates neural migration in different patterns. One scenario involves a Reelin signaling related mechanism, and another possible mechanism involves the regulation of microtubule dynamics (Ables et al. 2011). A recent study further indicates that a Reelin-Notch crosstalk is required during cortical neuron migration (Hashimoto-Torii et al. 2008). In addition, endocytosis is also involved in neocortical migration. Clathrin–mediated endocytosis (CME) takes effect in regulating substrate detachment to enable soma translocation in migrating neurons by modulating the subcellular distribution of cell adhesion proteins at the neuroblast surface (Shieh et al. 2011).
Actin filaments, one of the major components of cytoskeleton, are the structural element of the lamellipodia and filopodia as mentioned above. Thus, actin can modulate the leading process of migrating neurons. Molecules such as tropomyosin could regulate the polymerization and depolymerization of actin and thus influence neuronal migration. In addition, actin also affects nuclear movement in many kinds of cells including the developing neurons (Luxton et al. 2011). But how these molecules mediate the contact between actin and the nucleus remains still unclear. A very important motor protein which could bind to F-actin named myosin II has been shown to influence neuronal migration (Vallee et al. 2009). Researchers who used blebbistatin, the inhibitor of myosin II, have found that actomyosin filaments are accumulated at the rear of nucleus (Bellion et al. 2005; Vallee et al. 2009). These results showed that myosin II may push nucleus to move forward and control the nuclear movement. Also, diabetes mellitus condition influences the brain function by modifying expression of myosin II (Calabria et al. 2011). Recent studies further indicate that a protein complex named linker of nucleoskeleton and cytoskeleton (LINC) complex, which is composed by nesprin proteins and SUN proteins, could connect with both actin and nucleus (Luxton et al. 2011). Maybe this complex constitutes the key that could explain how actin could drive nuclear movement.
Microtubules could affect nuclear movement by mutual antagonistic motors. Cytoplasmic dynein, a motor protein, moves toward the minus-end of microtubules (Vallee et al. 2009) and can be regulated by LIS1 (see also Chap. 1). LIS1 is encoded by the LIS1 gene and can cause Lissencephaly when mutated in human. Both LIS1 and cytoplasmic dynein can interact with the evolutionary conserved protein NDEL1 (Chansard et al. 2011). NDEL1 can recruit LIS1 and dynein to the nuclear envelope (Chansard et al. 2011; Vallee et al. 2009). This microtubule-LIS1-cytoplasmic dynein complex could link to SUN-nesprin complex and accelerate nuclear movement. In certain instances, neurons need to retard this movement. A slow motor molecule called kinesin-5 is thought to be the brake of nuclear movement (Falnikar et al. 2011). Thus, several motor proteins modulate the nuclear movement.
4 Clinical Perspectives – Neuronal Migration Diseases
Neuronal migration is a critical process in cortical development and the defects in neuronal migration can lead to devastating brain diseases. For example, patients suffering from Lissencephaly harbor cortical malformations resulting from defective radial neuron migration and failure of cortical fold formation (see also Chaps. 1 and 5). As discussed earlier, the loss function of LIS1 is a major cause of lissencephaly, leading to abnormal nuclear translocation during neuronal migration and hence impairing brain gyrus formation (LaMonica et al. 2012; Wu and Wang 2012). Mutation of Reelin, a molecule that plays a critical role in neuronal migration regulation during cortical development as discussed above, also lead to lissencephaly (Hong et al. 2000). In addition, various mutation of Reelin have also been reported in neuropsychiatric disorders, including schizophrenia, bipolar disorder and autism, and neurodegenerative disease, like Alzheimer’s disease (AD) (Botella-Lopez et al. 2006; Chin et al. 2007; Kelemenova et al. 2010; Nahin et al. 1991; Persico et al. 2001; Rogers et al. 2011).
5 Neuronal Migration and Therapeutic Strategies for Brain Disorders
Although not many neurons migrate in the adult brain, cellular migration research is still important with respect to stem cell therapies for some brain diseases. Neural stem cells have been considered as potential and effective methods for neurodegenerative diseases and CNS injury due to their great abilities to proliferate and differentiate into specific terminal cell types. There are numerous studies using animal models, such as rodents and primates, showing that transplantation of neural stem cells could be effective to some extent in the treatments of Parkinson’s disease (PD), Amyotrophic lateral sclerosis (ALS), Alzheimer’s disease (AD) and stroke (Blurton-Jones et al. 2009; Darsalia et al. 2007; Kelly et al. 2004, 2005; Kim et al. 2006; Redmond et al. 2007; Takagi et al. 2005). Defining the experimental conditions to promote stem cells migration to the desired regions is one of the most important challenges that need to be overcome for the success of neurological diseases therapies. One can expect that efficient migration to the desired destinations would favor appropriate neural stem cell differentiation, and integration into neuronal circuits. Indeed, the regulation of stem cell migration involves the contribution of ECM components and intracellular signals, including some members of the cytokine family. Stem cell factor (SCF), originally was characterized as a molecule playing an important role in the regulation of hematopoiesis, spermatogenesis and melanogenesis during development. Recent further studies have revealed that both SCF messengers and proteins are highly expressed in neurons at injured sites in the brain. SCF signals promote neural stem cell migration to lesions in vitro and in vivo via the activation of its receptor c-kit (Jin et al. 2002; Sun et al. 2004). In a rodent model of Huntington’s disease, the activation of SCF and c-kit signaling pathways are required for transplanted NSCs to migrate to the diseased striatum (Bantubungi et al. 2008).
In a rat model of stroke, human neural stem cells are able to migrate to the ischemic lesions and differentiate into neurons after transplantation (Darsalia et al. 2007; Kelly et al. 2004). Recently, further studies in stem cell therapy for stroke have demonstrated that pre-differentiated brain-derived adult human progenitor cells migrate more efficiently to the stroke damaged area after transplantation in vivo, due to the signaling of chemokine receptor 4 (CXCR4) and its ligand, stromal cell-derived factor-1α (SDF-1α), which are both highly expressed in pre-differentiated cells and ischemic regions respectively (Olstorn et al. 2011).
With the rapid emergence of sophisticated molecular biological cell-label techniques and powerful cell-tracing imaging systems, future studies on the regulation of neural migration in embryonic (developing) and adult (mature) CNS will not only reveal the fundamental underlying basis of pathological neurological disorders, but also open new avenues to find prospective candidate drugs and therapeutic strategies for CNS diseases and injury.
References
Ables JL, Breunig JJ, Eisch AJ, Rakic P (2011) Not(ch) just development: Notch signalling in the adult brain. Nat Rev Neurosci 12(5):269–283. doi:10.1038/nrn3024 PubMedCrossRef
Ang ES Jr, Haydar TF, Gluncic V, Rakic P (2003) Four-dimensional migratory coordinates of GABAergic interneurons in the developing mouse cortex. J Neurosci 23(13):5805–5815PubMed
Asensio VC, Campbell IL (1999) Chemokines in the CNS: plurifunctional mediators in diverse states. Trends Neurosci 22(11):504–512PubMedCrossRef
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








