Vascular Injuries of the Head and Neck
This chapter will focus on the more commonly seen traumatic injuries to the great vessels of the neck and intracranial circulation, including dissections, pseudoaneurysms, and lacerations. The etiology, diagnosis, and management of these injuries will be discussed as well.
Arterial Dissection
Etiology
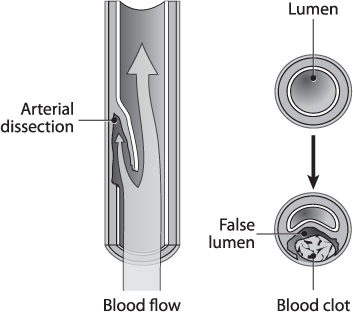
Traumatic arterial dissections occur as a result of shearing forces between the layer of luminal endothelial cells (intima) and the smooth muscle layer deep to it (tunica media). This tear results in a tissue flap within the arterial lumen, creating a false passage for blood to propagate into the subintimal space (Fig. 19.1). Enlargement of this false lumen can narrow or obstruct the true arterial lumen, resulting in diminished blood flow at, and distal to, the site of injury. The torn endothelium and exposed basement membrane are thrombogenic, and platelet adhesion to these damaged layers places the patient at risk for cerebral hypoperfusion and thromboembolic events. Additionally, a pseudoaneurysm can form at the dissection site secondary to further weakening of the arterial wall.
Arterial dissections are more commonly seen in the cervical segment of the external carotid (ECA), internal carotid (ICA), and vertebral (VA) arteries than in the intracranial segments.1,2 Spontaneous dissections are much less common than traumatic ones and are often associated with connective tissue disorders (Table 19.1). If the dissection spreads into the intracranial space, where the arterial wall is thinner, subarachnoid hemorrhage can result.3 Rapid rotation or extension of the head or neck, a fracture through the skull base or cervical vertebrae, and facial trauma are the most common causes of traumatic arterial dissections of the ICA and VA; occasionally, however, there is no apparent inciting event.
Diagnosis
Extracranial carotid artery (CA) dissections most commonly occur 2–3 cm above the common carotid artery (CCA) bifurcation and are seen in up to 0.45% of blunt neck trauma patients, with an associated mortality of 40%.4 Symptomatic vertebral artery injury is diagnosed in up to 1% of blunt neck trauma cases. Screening studies have shown that the incidence of asymptomatic vertebral artery injury from blunt trauma in patients with mid-cervical spine fractures or subluxations is as high as 46%. Many arterial dissections go undiagnosed because they are often asymptomatic. However, the onset of neurological deficits from CCA, ICA, and VA dissections can occur any time from immediately after the injury to days later; most occur within 24 hours. Carotid dissections may present with ipsilateral neck pain over the carotid artery (carotidynia) or neurological deficits (Table 19.2).
Fig. 19.1 Anatomy of an arterial dissection.
Vertebral artery dissections can present with pain in the posterior neck and symptoms of posterior circulation ischemia (Table 19.3). These dissections are more commonly seen in the first segment of the VA (cervical segment) at its origin from the subclavian artery and the third segment as the VA exits C2. Ten percent of these dissections extend intracranially, with a risk of subarachnoid hemorrhage (SAH) associated with high morbidity.
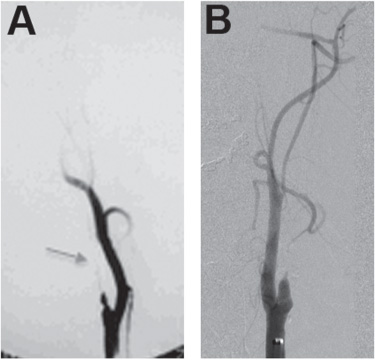
Angiography remains the gold standard for detecting arterial dissections in both the neck and skull base, although CT angiograms (CTA) are being utilized in an increasing number of cases, particularly in the setting of trauma, where CTA of the head and neck can be rapidly obtained as part of the trauma evaluation. The characteristic angiographic appearance of an arterial dissection is a smooth tapering of the true vessel lumen called a “string sign,” (Fig. 19.2A) or in severe dissections, an abrupt cessation of contrast filling representing an occlusion (Fig. 19.2B).
Ultrasonography is a useful tool for rapidly evaluating a patient for cervical arterial dissections, particularly for the carotid artery, since it does not lie in a bony canal like the VA. Arterial lumen narrowing from a dissection is suggested by abnormally high blood flow velocities on ultrasound. However, occasionally a dissection can be difficult to differentiate from hypoplasia with ultrasonography, in which case magnetic resonance angiography (MRA), CTA, or catheter-based angiography (CBA) is more helpful because these modalities can demonstrate the intimal flap. Ultrasonography is less useful for detecting intracranial arterial dissections because the bone density interferes with the sonar waves, resulting in poorer quality images. In these situations, CBA, CTA, or MRA is preferred.
Table 19.1 Connective Tissue Disorders
Ehlers-Danlos syndrome |
Polycystic kidney disease |
Marfan syndrome |
Loeys-Dietz syndrome |
Table 19.2 Anterior Circulation Exam Findings in Patients with Carotid Dissection
Contralateral hemiparesis |
Hemisensory deficits |
Aphasias |
Agnosias |
Hemineglect |
Ipsilateral Horner syndrome |
Table 19.3 Posterior Circulation Exam Findings in Patients with Vertebral Artery Dissection
Cortical blindness |
Vertigo |
Vomiting |
Cranial nerve deficits |
Coma or death |
Fig. 19.2 Angiographic appearance of an arterial dissection.
Evidence-Based Medicine
To date, there have been no randomized controlled trials evaluating arterial dissections. At the time of this writing, the Cervical Artery Dissection in Stroke Study (CADISS) has been initiated.5 We look forward to the results of this study, which is assessing medical treatment of cervical arterial dissections.
Stroke Rates
Cervical dissection, whether spontaneous or traumatic, accounts for approximately 2% of all ischemic strokes.6
Management
Conservative Therapy
The natural history of dissection is somewhat unclear. Asymptomatic carotid artery and VA dissections frequently heal with anticoagulation or antiplatelet agents alone. However, for CA or VA dissections, no randomized clinical trials have compared antiplatelet or anticoagulant agents to each other or to no treatment at all.7,8 Yet, during the healing period, the patient is at risk for two potential complications: (1) extension of the arterial dissection, and (2) thromboembolic infarcts. Some studies have shown that 50–90% of these tears will heal completely.6 Others report that the rate of spontaneous healing of the dissection is much lower and that early intervention is imperative.9 Morbidity from stroke ranges between 8% and 80%.6,9
Despite the absence of data supporting or refuting the use of antiplatelet and anticoagulant agents, they are commonly used during the healing phase (generally up to six months) to lower the risk of thromboembolic events. Once an arterial dissection is diagnosed, urgent treatment with intravenous heparin and oral warfarin (Coumadin) or antiplatelet agents should be initiated to prevent thromboembolic infarcts after all other potential sources of bleeding have been identified and treated. Intracranial hemorrhage on CT is a contraindication to anticoagulation. In the absence of intracranial hemorrhage, intravenous heparinization (with the goal of a partial thromboplastin time [PTT]/control ratio of 2.0–2.5) and oral warfarin (Coumadin) should be initiated urgently until the International Normalized Ratio (INR) is 2.0–2.5, at which time the heparin can be stopped; initial use of heparin reverses the early hypercoagulability seen with warfarin. If antiplatelet agents are used, heparin does not necessarily need to be started in conjunction, but this is also debatable and left to individual physician preferences.
Treatment with antiplatelet or anticoagulant agents should be continued for a minimum of six months, at which time imaging should be performed to assess whether or not the luminal stenosis from the dissection is stable or improved, suggesting that the intimal tear has healed. If the dissection has healed, the agents can be discontinued; if the stenosis is worse or if the patient becomes symptomatic, then a stent is recommended for definitive treatment.9
It is important to recognize the small group of patients who complain of vague or intermittent neurological symptoms that cannot clearly be attributed to an arterial dissection. In such circumstances, there are two studies that we find helpful: CT or MR perfusion study and continuous transcranial Doppler (cTCD). Perfusion studies are useful when deciding whether or not the dissection is causing cerebral hypoperfusion.
Surgery
Open surgical treatment of arterial dissections traditionally meant arteriotomy with suturing of the intimal flap against the arterial wall or arterial sacrifice, with or without bypass. The latter surgical method has typically been more successful because the friability of the vessel wall and the length of the dissection usually preclude tacking the intimal flap to the arterial wall as a viable option. Vessel sacrifice can be done via multiple methods, although the most common in the endovascular era is with coil embolization or endovascular arterial occlusion devices. Sacrifice can be done by surgical ligation as well. A balloon occlusion test should be performed first to make sure the patient would tolerate arterial occlusion. If the patient fails the temporary balloon occlusion test, an intracranial to extracranial arterial bypass by a cerebrovascular neurosurgeon is recommended prior to vessel sacrifice.
Pseudoaneurysms
Extracranial Pseudoaneurysms
Etiology and Diagnosis
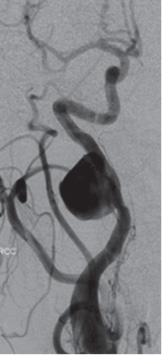
When there is injury to both the arterial intima and tunica media, a false channel forms between the tunica media and outer adventitia that fills with blood. With only a thin layer of adventitia between the tunica media and surrounding soft tissue of the neck, the false lumen expands and creates a pseudoaneurysm (Fig. 19.3). The injury occurs when the neck is hyperextended, causing stretching of the ICA and VA that is similar to that of dissections. Unlike dissections, pseudoaneurysms cannot form in arterial segments that are securely encased in bone, such as the petrous segment of the ICA, since there is no room for the adventitia to expand.
Extracranial pseudoaneurysms are more commonly seen in patients with injury to the neck or skull base after penetrating trauma or complications of otolaryngological surgery. Patients who present with pseudoaneurysms after blunt injury to the neck typically have pain localized to the general area of the injury, a neck mass, or oropharyngeal compromise in the case of larger cervical carotid pseudoaneurysms.
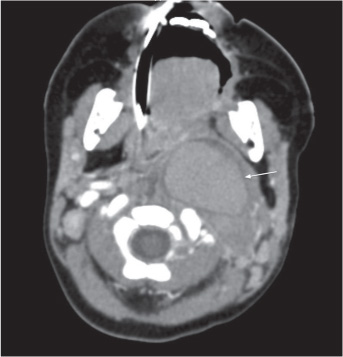
Arterial duplex ultrasound can be used to detect cervical carotid and vertebral artery pseudoaneurysms, the distinguishing feature being an extraluminal mass with slow blood flow. Pseudoaneurysms of the CA and VA appear as a mass on CT and MRI that is consistent with hematoma in the pseudoaneurysm (Fig. 19.4). The sensitivity of CTA in diagnosing pseudoaneurysms is 80%. CBA demonstrates flow through the lumen of the parent artery, usually without luminal narrowing. The injured segment of the parent artery can be identified angiographically as it fills the pseudoaneurysm pouch.
Fig. 19.3 Pseudoaneurysm.
Fig. 19.4 Noninvasive CT-angiography of a pseudoaneurysm.
Management
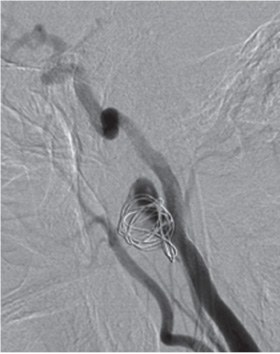
Small, asymptomatic, extracranial cervical carotid pseudoaneurysms can be observed with serial imaging studies long term. If the pseudoaneurysm expands or becomes symptomatic (thromboembolic events or soft tissue compression of the airway and oropharynx), then stent placement, stent-coil embolization, or vessel sacrifice may be necessary (Fig. 19.5).
Intracranial Pseudoaneurysms
Etiology and Diagnosis
Intracranial pseudoaneurysms are rare and unstable injuries that are associated with a very high risk of rupture if they are left untreated. The mortality rate is reported as high as 50% from re-rupture of untreated lesions, particularly within the first three weeks after diagnosis.10–12 Pseudoaneurysms account for less than 1% of all intracranial aneurysms, with an estimated incidence of 0.15–0.40% of all diagnosed intracranial aneurysms.11 A few relatively large case series are reported, evaluating up to 31 patients with cerebral angiography after head trauma, and reporting an associated incidence of traumatic intracranial pseudoaneurysms anywhere from 3.2% to 42%.10
Fig. 19.5 Treatment of a pseudoaneurysm.
The majority of intracranial pseudoaneurysms occur secondary to iatrogenic injury, penetrating trauma, infectious origin, or rupture of intracranial aneurysms into the brain parenchyma. More rarely, intracranial pseudoaneurysms can occur as a result of blunt trauma, particularly when skull base fractures involve the carotid canal; direct surgical exposure; and microwire perforation during endovascular procedures.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree