Fig. 7.1
Immunohistochemical localization of islet cell hormones in normal islets. (a) Insulin localization in approximately 60–70 % of islet cells. (b) Glucagon localization in the periphery of the islets in approximately 15–20 % of islet cells. (c) Somatostatin localization in approximately 5–10 % of islet cells. (d) Human pancreatic polypeptide is present in 2–5 % of islet cells in the body of the pancreas
7.1.1 Transcription Factors in Pancreatic Neuroendocrine Tissues
A large number of transcription factors (TFs) that are critical for development of the normal pancreas have been identified in PanNETs. The four principal TFs include islet 1 gene product (ISL1) (Fig. 7.2), pancreatico-duodenal homeobox 1 gene product (PDX1), neurogenin 3 gene product (NGN3), and CDX2 homeobox gene product (CDX2) [7–14]. Studies have been done to correlate the PanNET hormonal profile with specific TFs. Insulin-positive tumors usually express ISL1. In a recent study, 90 % of primary PanNETs were positive for ISL1, while only 76 % of metastatic PanNETs were positive indicating loss of expression in the malignant tumors [12]. Glucagon-positive tumors express ISL1 and are usually negative for most of the other principal TFs. Gastrin-producing tumors express PDX1, ISL1, and NGN3. CDX2 is often found in gastrin-producing cells. Somatostatin-producing cells often express ISL1, PDX1, and NGN3. There is usually not an association between TF patterns of expression and PanNET features such as grade, size, location, presence of metastases, and functional activity. Studies in experimental animals, especially knockout mice, have shown the functional role of many TFs in pancreatic development [2]. For example, animals lacking NGN3 fail to develop endocrine cells. Other TFs like PAX4 are involved in the early phases of development of endocrine lineages, while others like PAX6 are involved in the maturation of committed endocrine cells [2].
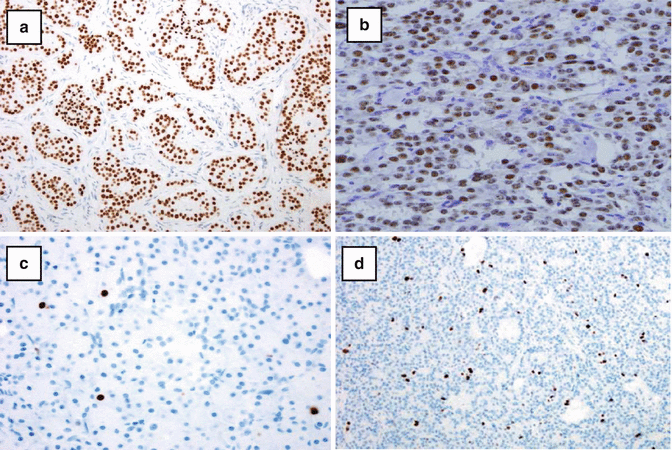

Fig. 7.2
(a) Localization of ISL1 in an insulin-producing pancreatic neuroendocrine tumor. (b) Localization of INSM1 in an insulin-producing pancreatic neuroendocrine tumor. (c) Immunostaining for Ki-67 in a grade 1 pancreatic neuroendocrine tumor with a labeling index of 2 %. (d) Immunostaining for Ki-67 in a grade 2 pancreatic neuroendocrine tumor with a labeling index of 10 %
INSM1 is a zinc-finger TF expressed transiently in developing embryonic and regenerating adult neuroepithelial tissue. INSM1 was originally isolated from human insulinoma and glucagonoma tissues, from which it took its name (insulinoma-associated protein 1). INSM1 coordinates differentiation of neuroendocrine and neuroepithelial cells with termination of cell division and is thought to have critical regulatory roles during neuroendocrine differentiation. It has been detected in many neuroendocrine tissues in rodents and other animals. In human adult tissues, INSM1 has been identified in multiple tumors of NE or of neuroepithelial origin. Recent studies have shown that INSM1 is widely distributed in normal islet cells, and it has been detected in many different types of islet cell tumors (Fig. 7.2b) [15, 16].
7.1.2 Ki-67 in Pancreatic Neuroendocrine Neoplasms
Immunohistochemical staining for Ki-67 (Figs. 7.1c and 7.2d) along with mitotic counts is critical for the grading of PanNETs [17–19]. Grade I (G1) tumors usually have a Ki-67 index of less than 3 % and less than two mitoses per ten high power fields (HPF). G2 tumors have a Ki-67 index of 3–20 % or 2–20 mitoses per 10 HPF. G3 or poorly differentiated pancreatic endocrine carcinomas have a Ki-67 index greater than 20 % and >20 mitoses per 10 HPF [19]. It is recommended that at least 500 tumor cells be counted in tumor hot spots for calculation of the Ki-67 index. If there is discordance between the Ki-67 index and mitotic count, the higher grade should be used (Table 7.1).
Table 7.1
Immunohistochemical markers for islet cells and pancreatic neuroendocrine tumors
Secretory granule-associated proteins |
Chromogranins A and B, secretogranins I and II and 7B2 |
Prohormone convertases PC1/3 and PC2 |
Membrane-related proteins |
Synaptophysin |
Synaptic vesicle proteins 2 |
Synaptotagmins |
Vesicular monoamine transporters |
Vesicle-associated membrane protein |
Cytoplasmic proteins |
CD56 |
Neuron-specific enolase |
PGP 9.5 |
Proliferation markers |
Ki-67 |
Cell membrane -associated protein |
Somatostatin receptors |
One potential source of variation between laboratories with the Ki-67 index is the method used to obtain and to analyze the data. There are a limited number of monoclonal antibodies (MIB1) used for immunostaining. Most of these produce highly reproducible results—especially with automated staining—so the method of immunostaining is usually not a significant problem. Visually estimating the percentage of positive cells with the “eyeballing” method is usually not reproducible [17]; in contrast, automated counting is more reliable, but may show some operator variability depending on the experience of the operator and the quality of the software used in the analysis. Tumor heterogeneity and subjectivity in determining the hot spots for analyses probably contribute to variation in proliferation indices between different laboratories.
7.1.3 Broad-Spectrum Immunohistochemical Staining of PanNETs
PanNETs are characterized by the presence of secretory granules and membrane-bound vesicles. The chromogranins, including chromogranin A and chromogranin B, and secretogranins are present in secretory granules. Chromogranin A is widely used to characterize PanNETs (Fig. 7.3a) [20–23]. It is a very specific neuroendocrine marker, but is not as sensitive as some other markers. The sensitivity is most likely related to the number of secretory granules in the neoplasm. Synaptophysin (Fig. 7.3b) which is associated with cytoplasmic membrane vesicles is highly sensitive [24], but not as specific as chromogranin A, since some non-neuroendocrine tumors, such as the adrenal cortex, also express synaptophysin.
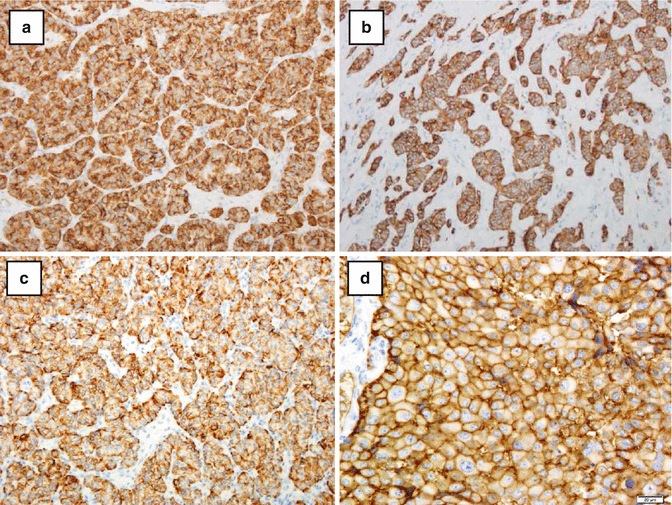

Fig. 7.3
(a) Chromogranin A expression in a pancreatic neuroendocrine tumor. There is diffuse staining for chromogranin A which labels the protein in the dense core secretory granules. (b) Localization of synaptophysin in membrane-related vesicles in a pancreatic endocrine tumor showing diffuse cytoplasmic staining. (c) Localization of cytokeratin 19 in an insulin-producing pancreatic endocrine tumor. (d) Localization of somatostatin receptor protein type 2 in a pancreatic endocrine carcinoma with a Ki-67 labeling index of 2 l% showing distinct cytoplasmic membrane staining (Courtesy of Dr RY Osamura)
CD56, a broad-spectrum neuroendocrine marker, directed against cell adhesion molecules, is the least specific of these three principal neuroendocrine markers, but is used in practice when the differential diagnosis includes a neuroendocrine tumor and chromogranin A and synaptophysin are negative [22]. Protein Gene Product 9.5 (PGP 9.5) belongs to the carboxy-terminal hydrolase family and is involved in the degradation of cytosolic and nuclear proteins through an ATP- and ubiquitin-dependent mechanism. It has a role in the regulation of the cell cycle. Although it is expressed in the normal pancreatic islets and in PanNETs, PGP9.5 is also expressed by the exocrine pancreas, so it is not a specific marker for PanNETs [22]. Neuron-specific enolase (NSE) was historically the first broad-spectrum neuroendocrine marker, but because of the relative lack of specificity is not used frequently in many diagnostic pathology laboratories [25]. Other broad-spectrum markers that may also be positive in PanNETs include prohormone convertases (PCs), PC2 and PC1/PC3 [22]. Additional membrane-related proteins in addition to synaptophysin such as synaptic vesicle protein, synaptotagmins, vesicular monoamine transporters, and vesicle-associated membrane proteins are also potentially useful broad-spectrum neuroendocrine markers, but they are not widely used [22].
7.1.4 Miscellaneous Broad-Spectrum Markers
Cytokeratin 19 (CK19) may be expressed by some PanNETs (Fig. 7.3c). Some reports indicated that CK 19 was a marker for some aggressive PanNETs [26, 27]. CD117 (C-Kit) has been reported to be a marker with prognostic significance in PanNETs. It was found to be an independent prognostic marker in more aggressive PanNETs [28].
The gene products of the death domain-associated proteins DAXX and ATR-X which encode proteins involved in chromatin remodeling have been detected in 40 % of PanNETs. The expression of these markers has been associated with activation of alternative lengthening of telomeres [29]. DAXX and ATR-X protein loss have been associated with tumor stage and metastasis, reduced time of relapse-free survival, and decreased time of tumor-associated survival. Marinoni et al. have suggested that mutations of DAXX and ATRX along with chromosome instability have been associated with PanNET progression [29].
7.2 Somatostatin Receptors
Somatostatin receptors are members of the seven-transmembrane G-protein coupled receptor superfamily which are widely distributed throughout the body. There are five subtypes of somatostatin receptors (SSTs) [30, 31]. The main function of somatostatin is the inhibition of secretion of various hormones. This function is mediated through SSTs 2. Suppression of cell growth is carried out by SSTS 1, 2, and 5. The presence of SSTs indicates whether tumors will respond to somatostatin analog therapy, which usually acts on SST subtypes 2, 3, and 5. The presence of the SST subtypes has been studied by receptor autoradiography, in situ hybridization, polymerase chain reactions, and immunohistochemistry (Fig. 7.3d) with subtype-specific antibodies [30, 31]. All five SSTs have been identified in the normal islets. SST 5 was not present in normal PP cells. The expression may vary with different types of PanNETs. For example, insulinomas may not express some SST subtypes. This correlates with the observation that around 20–50 % of insulinomas are unresponsive to somatostatin analog treatment [2]. This emphasizes the importance of determining the presence of and specific subtypes of SSTs before treatment of pancreatic PanNETs with somatostatin analog therapy (Table 7.2).
Table 7.2
Hormones produced by pancreatic neuroendocrine tumors
Functioning tumors |
Insulinoma |
Glucagonoma |
Somatostatinoma |
Vasoactive intestinal polypeptide (VIPoma) |
Gastrinoma |
Enterochromaffin-cell tumors (serotonin) |
Nonfunctioning tumors |
Pancreatic polypeptide |
Ectopic hormone production–functioning tumors |
Growth hormone releasing hormone (GHRH) |
Growth hormone (GH) |
Ghrelin |
Corticotropin-releasing hormone (CRH) |
Adrenocorticotropin hormone (ACTH) |
Parathyroid hormone (PTH) |
Parathyroid hormone-related peptide (PTHrp) |
Ectopic hormone–nonfunctioning tumors |
Calcitonin |
7.2.1 Hormone Producing Functioning PanNETs
7.2.1.1 Insulinomas
Insulinomas are the most common functioning PanNETs. They occur most frequently between 30 and 60 years of age [32–34]. Insulinomas are generally small with an average diameter of 1.5 cm. Patients with insulinomas usually present with symptoms including hypoglycemia, headaches, weakness, dizziness incoherence, convulsions, or coma. Insulinomas occur most frequently in the body and tail of the pancreas as solitary benign tumors which are usually treated by surgical resection or enucleation. They may show different patterns of growth including trabecular-gyriform, lobular, and solid patterns. Amyloid (composed of amylin protein) may be present in insulin-producing islet cells and in insulinomas (Fig. 7.4a). After Congo Red staining and examination with polarized light, amyloid in insulinoma shows the characteristic green birefringence. Insulin immunostaining is present in almost all insulinomas (Fig. 7.4b). Many tumors are also positive for proinsulin and for amylin. About half of insulinomas are multihormonal with cells also positive for glucagon, somatostatin, gastrin, and PP. Most insulinomas are benign (90–95 %). Malignant insulinomas are diagnosed by metastatic or locally invasive disease. Patients with malignant insulinomas have a median survival of only 4 years [2, 33].
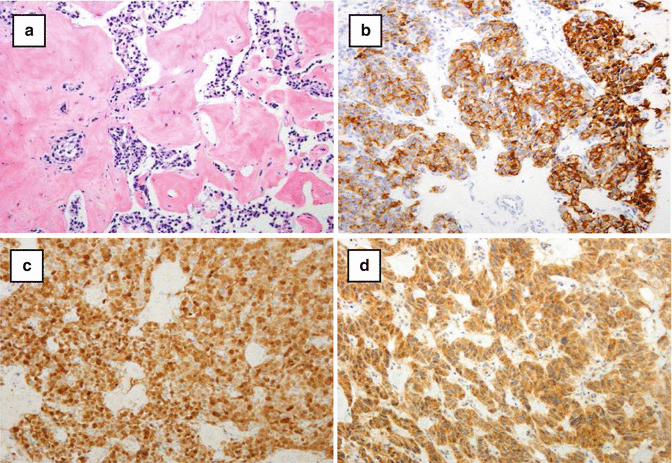

Fig. 7.4
(a) Congo red staining showing staining positive reaction for amyloid in an insulinoma. (b) Immunohistochemical staining for insulin showing diffuse positive staining in the tumor. (c) Pancreatic glucagonoma showing strong positive staining for glucagon in the tumor cells. (d) Pancreatic VIPoma showing strong positive staining in the tumor cells
7.2.1.2 Glucagonomas
Glucagonomas are usually well-differentiated PanNETs derived from the alpha cells. Patients may present with dermatitis (also known as necrolytic migratory erythema), stomatitis, diabetes, weight loss, and anemia [34–38]. They are uncommon even for pancreatic endocrine tumors and represent about 8 % of functioning PanNETs. Most tumors are solitary and are localized in the body and tail region with a mean diameter of around 7.6 cm. Most glucagonomas are malignant and patients may have local invasion or metastases to the liver, regional lymph nodes, bone, lungs, and adrenals. Glucagonomas are characterized by a trabecular and diffuse pattern of growth. Vascular and perineural invasion are common. Immunohistochemical stains are positive for glucagon (Fig. 7.4c) and for peptides derived from proglucagon, such as glicentin, and glucagon-like peptides. Glucagonomas may also express other pancreatic islet cell hormones. About 80 % of glucagonomas are malignant and approximately 70 % of patients have metastases at the time of diagnosis. Like many PanNETs, glucagonomas grow slowly and patients may survive for many years after surgical resection [2, 33].
7.2.1.3 Somatostatinomas
Somatostatinomas are PanNETs associated with the somatostatinoma syndrome which includes diabetes mellitus, cholelithiasis, diarrhea with or without steatorrhea, weight loss, hypochlorhydria, and anemia [2, 33]. The tumors are commonly located in the head of the pancreas, but may arise anywhere in the pancreas. Somatostatinomas are more common in the duodenum than in the pancreas. They are generally large tumors ranging in average diameter from 5 to 6 cm. The histological spectrum of somatostatinomas may range from trabecular and acinar patterns to solid sheets of tumor cells growing in a diffuse manner. Tumors frequently show angioinvasion and perineural invasion. Psammoma bodies, which are frequently found in duodenal tumors, are rare in pancreatic tumors. Immunohistochemical staining for somatostatin can be quite variable. Immunoreactivity for other hormones including gastrin and calcitonin may be present [2].
7.2.1.4 Vasoactive Intestinal Polypeptide-Producing Tumors (VIPomas)
VIPomas occur mainly in the pancreas and are associated with the Verner-Morrison syndrome which includes watery diarrhea, hypokalemia, and achlorhydria (WHDA) due to the secretion of vasoactive intestinal polypeptide (VIP), peptide histidine methionine (PHM), and related hormone-like substances [39]. VIPomas are rare pancreatic endocrine tumors. Most VIPomas are solitary tumors, and they are most frequent in the tail of the pancreas. Tumors have an average size of 5 cm. The metastatic rate ranges from 40 to 70 % [2]. The tumors may grow as solid, trabecular, or tubulo-acinar patterns and microcysts may be present. Vascular and perineural invasion is a common finding. Immunoreactivity for VIP is usually detected (Fig. 7.4d), and the tumors may also be positive for PHM, PP, or somatostatin. Around 80 % of VIPomas may be metastatic at the time of diagnosis. If surgery is not effective, treatment with long-acting somatostatin analogs may help to control WHDA symptoms in some patients [2, 33].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







