Fig. 22.1
Approved and not approved therapies in NENs. IFN interferon, PRRT peptide radioreceptor therapy
22.2 General Approach
Therapeutic approach can change according to the functioning/nonfunctioning status of the neoplasm. In functioning PNENs, the therapeutic priority is the syndrome control. Diazoxide in insulinomas and proton pump inhibitors (PPIs) in gastrinomas represent two medical therapies to be considered upfront. Somatostatin analogs (SSAs) are indicated in VIPoma, in glucagonoma, rarely in gastrinoma and insulinoma, and in very rare functioning PNENs, like ACTHomas, carcinoid syndrome-related PNENs, or hypercalcemia-related PNENs (PTHrp-omas) [1].
As for antiproliferative treatment, therapeutic approach varies according to the grade of malignancy and clinical behavior of the neoplasm. More in general for advanced fast-growing PNENs, chemotherapy is usually the only option, whereas for advanced slow-growing PNENs, many other options can be considered (Fig. 22.2).
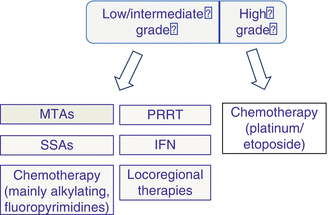

Fig. 22.2
PNENs: therapeutic options related to the grade of malignancy. MTAs molecular-targeted agents, SSAs somatostatin analogs, IFN interferon, PRRT peptide radioreceptor therapy
Therefore, it is crucial differentiating between the two categories. An important help comes from the 2010 pathology classification that distinguishes G1/G2 from G3 PNENs [2]; in other words, G1/G2 would correspond to low/intermediate grade of malignancy and G3 to the high grade. The first group is also the most numerous that means that in our clinical practice, it is most common to run into G1/G2 than into G3 PNENs. On the other hand, Ki-67 and/or mitotic index (MI) and/or tumor morphology is not enough to be sure to have understood the clinical behavior of the neoplasm or its response to therapy. Other factors, including patient’s and neoplasm’s characteristics, are usually necessary to better understand the best therapy to be proposed. Except for a minority of cases where the clinical picture of the tumor and/or patient is particularly severe or rapidly worsening, the vast majority of cases can be faced applying an algorithm including confirmation of diagnosis, definition of prognostic characteristics, performing an adequate staging, and discussing therapy and therapeutic strategy within a multidisciplinary dedicated team (Table 22.1). It is essential to be sure that the patient really has a “pure” PNEN (neither a mixed tumor nor an adenocarcinoma with neuroendocrine differentiation); sometimes, it requires a second opinion by a pathologist particularly skilled in NEN.
Table 22.1
Algorithm for diagnosis, characterization, and prognosis estimate of the disease
Steps | Key point | Specialist |
|---|---|---|
Right diagnosis | “Pure” PNEN | Pathologist |
Histological prognostic factors | Tumor differentiation, Ki-67/MI | Pathologist |
Morphological imaging | CT scan/MRI | Radiologist |
Functional imaging | SRS/68Ga-DOTA-octreotide PET-CT | Nuclear medicine expert |
18FDG PET-CT | ||
Clinical picture | Syndrome/symptoms/P.S. | Clinician |
22.3 Fast-Growing PNENs
This category is represented mainly by high-grade PNENs, which are named carcinomas (PNECs) according to the 2010 WHO classification. Basically for this category, chemotherapy represents the only therapeutic option. Historically in advanced stage NECs, platinum/etoposide has been the most used chemotherapy; it represents a sort of standard of care, although it is based on old and small-size studies [3–5]. It is based on the assumption that a NEC is clinically similar to the small cell lung cancer (SCLC). However, increasing clinical knowledge seems to support different categories of PNECs.
Probably, a small cell neuroendocrine carcinoma (SCNEC), with a high Ki-67 (>70 %), will be directly related with a fast growth, and in that case, an approach similar to that of the SCLC is shared within the clinical community. However, some data reported a different response rate (RR) and survival between patients with gastroenteropancreatic (GEP) NECs with >55 % and <55 % Ki-67 [6]. Furthermore, some authors reported that among patients with GEP NECs (all with >20 % Ki-67), those with a well-moderately differentiated tumor morphology did not respond to platinum-based chemotherapy although their survival was better compared with that of patients with poorly differentiated tumor morphology [7]. Therefore, in the “low” Ki-67 PNECs, chemotherapies different from platinum/etoposide could be considered, like alkylating-based regimens, and even biological therapies, like everolimus (EVE) or sunitinib (SUN) or peptide receptor radionuclide therapy (PRRT), could be discussed for “well-differentiated PNEC.”
22.4 Slow-Growing PNENs
This category is represented mainly by low-/intermediate-grade PNENs, which are named tumors (PNETs) according to the 2010 WHO classification. It is known that this category is clinically heterogenous. It includes different clinical histories, from advanced PNETs radiologically stable even for months without any antitumor therapy to PNETS with a pronounced radiological progression of disease within 2–3 months. Usually the former group has a very low Ki-67 (<5 %), well-differentiated tumor morphology, and high functional expression of somatostatin receptors (sstrs) at somatostatin receptor scintigraphy (SRS) or 68gallium-DOTA-octreotide positron emission tomography-computed tomography (68Ga-DOTA-octreotide PET-CT), with a patient in a very good clinical condition.
A number of different nonsurgical therapies can be considered in PNETs, including SSAs, interferon (IFN), several chemotherapeutics, molecular-targeted agents (MTAs), peptide receptor radionuclide therapy (PRRT), and liver-directed interventional radiology procedures (Fig. 22.2).
The role of SSAs as antiproliferative agents in GEP NETs has been debated for several years. Based on retrospective and prospective evidence [8, 9], octreotide (OCT) or lanreotide (LAN) now can be effective in functioning or nonfunctioning PNETs, radiologically stable or slowly progressing, G1 or G2 (better if <10 % Ki-67), preferably with high sstr functional expression, independently from the tumor burden, with an asymptomatic patient.
In PNETs progressing on or after SSAs, EVE or SUN can be considered. These two MTAs were approved on the basis of results of two placebo-controlled phase 3 trials in patients with pancreatic well/moderately differentiated, advanced NENs, with a baseline radiological progression [10, 11] (Table 22.2).
Table 22.2




Randomized phase III trials with SSAs and MTAs in PNETs
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree







