12
Surgical Management of Petroclival Tumors
Petroclival meningiomas are among the most difficult cranial base lesions to treat. They are located deep in the skull base, have an unpredictable growth pattern, and often envelope multiple cranial nerves as well as important venous and arterial structures.1 Although the advent of skull base approaches has heralded a new era in the treatment of these lesions, the perioperative morbidity and mortality of such tumors remains significant.2–7 Several classification schemes have been proposed for petroclival meningiomas.8 Sekhar et al9 divide these lesions based on their site of origin and anatomic extension. Tumors are placed into three categories: those involving the upper clivus, those involving the middle clivus, and those involving the lower clivus. Based on the site of origin of the meningioma, the appropriate surgical approach may be chosen.10,11 This chapter presents four surgical options for removal of a petroclival tumor, along with their respective advantages and disadvantages.
♦ Posterior Transpetrosal Approach
Indication
The posterior transpetrosal approach is the most versatile approach to petroclival meningiomas. It is the preferred surgical option for large petroclival tumors, especially meningiomas. This procedure provides access to the entire clivus as well as to tumor extension into Meckel’s cave and the posterior cavernous sinus. This approach combines a mastoidectomy with a supra-and infratentorial craniotomy. The petrosectomy options include a retrolabyrinthine resection, which preserves hearing but provides a small avenue in which to work. The translabyrinthine technique sacrifices hearing and provides a somewhat larger working area. The transcochlear technique, which provides the largest working area via maximal petrous resection, sacrifices hearing and transposes the facial nerve.
The drawbacks of the posterior transpetrosal approach are the additional surgical time required for the exposure, possible injury to the vein of Labbé, the risk of temporal lobe contusion, and cerebrospinal fluid (CSF) leak. Prior to performing this approach, the entrance point of the vein of Labbé into the transverse sinus should be noted on the preoperative gadolinium-enhanced magnetic resonance imaging (MRI) scans. A more posterior entrance into the transverse sinus is favorable. If the vein of Labbé enters the dural sinus anteriorly at the transverse-sigmoid sinus junction, the exposure provided by this approach will be limited unless the sigmoid sinus can be divided, or the mastoidectomy is extended to include a translabyrinthine or transcochlear exposure. A preoperative arteriogram is sometimes needed to determine the location of these venous structures when their size and anatomic course is not apparent on the MRI. The patency of the torcula should be assessed before surgery to determine the relative importance of the ipsilateral sigmoid sinus–jugular vein drainage system.
Procedure
The patient is placed supine with the head turned 60 to 80 degrees to the side opposite the tumor. A roll is placed under the ipsilateral shoulder. The neck is slightly extended such that the temporal lobe will drop away from the middle fossa floor without much retraction. The abdomen is prepped for obtaining a fat graft to fill the mastoidectomy defect. Electrodes are placed for monitoring the facial nerve and sixth cranial nerve if desired. Auditory evoked brainstem recordings can also be performed. The incision should be varied depending on the anterior extent of the tumor. If the tumor extends significantly into the middle fossa, Meckel’s cave, or cavernous sinus, the incision should start in front of the tragus, extend superiorly behind the temporal hairline, and then curve posteriorly just below the origin of the temporalis muscle. The incision should then curve inferiorly across the region of the transverse sinus and then down behind the mastoid process (Fig. 12.1A). If the tumor does not extend into the middle fossa, Meckel’s cave, or cavernous sinus, then a C-shaped incision can be used (Fig. 12.1B). The skin flap is elevated anteriorly above the plane of the temporalis fascia and periosteum of the mastoid process. An anterior-based temporalis muscle–parietal periosteum flap is elevated. A mastoid periosteum–sternocleidomastoid muscle flap is elevated and retracted inferiorly (Fig. 12.2).
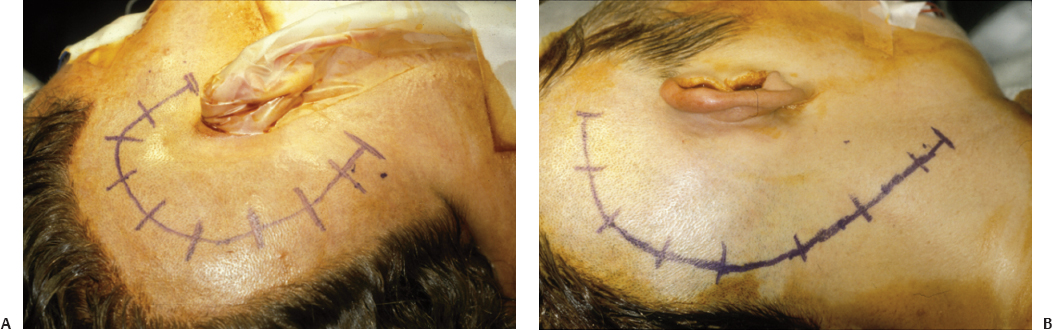
Fig. 12.1 Skin incisions for the posterior transpetrosal approach. (A) Incision used if the tumor extends into the middle fossa, Meckel’s cave, or cavernous sinus. (B) Incision used if the tumor is limited to the posterior fossa.
The sequence of bone work varies among surgeons. We prefer to start with the mastoidectomy. This enables us to unequivocally identify the location of the transverse sinus, which decreases the likelihood of this structure being injured while turning the craniotomy. The mastoidectomy must include removal of the bone along the dura of the middle and posterior fossa down to the level of the labyrinth, as well as the bone over the sigmoid sinus. Every millimeter of bone around the labyrinth should be removed, as this significantly improves the exposure of the base of the tumor. Exposing the distal sigmoid sinus just proximal to the jugular bulb is required to allow adequate posterior displacement of the sigmoid sinus. The dura is stripped off the undersurface of the convexity temporal, parietal, and occipital bones with a Penfield 3 dissector. The transverse sinus is stripped off the overlying calvaria. A trough of bone is removed with a cutting bur in the line of the planned craniotomy cut, over the posterior transverse sinus, exposing the dura both above and below the sinus. If the convexity dura is adherent to the skull, an additional trough-shaped bur hole is made over the superior temporal lobe. A one-piece supra-and infratentorial craniotomy is turned and the bone flap elevated (Fig. 12.3). Intravenous mannitol can be administered to aid in brain relaxation.
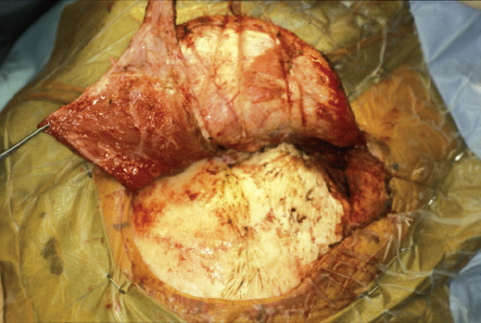
Fig. 12.2 Scalp opening with an anterior-based temporalis muscle flap and an inferior-based sternocleidomastoid periosteal flap.
The amount of temporal bone removed can be varied to improve the exposure as needed. There are several options, as described in the following subsections.
Retrolabyrinthine Petrosectomy
To preserve hearing, the integrity of the labyrinth is maintained. In addition to skeletonizing the lateral semicircular canal as already described, the posterior and superior canals are also skeletonized. Drilling is taken as far anterior as possible, exposing the maximal area of dura. This route provides good access to the cerebellopontine angle and lower clivus but does not provide direct visualization of the bony surface of the middle and upper rostral clivus.
Translabyrinthine Petrosectomy
The translabyrinthine approach, which sacrifices hearing, is accomplished via the same basic mastoidectomy already described, but the semicircular canals are also removed. The three canals are skeletonized and drilled away. Drilling then continues forward so that the posterior aspect of the internal auditory canal also can be skeletonized. The bone overlying the labyrinthine and mastoid segments of the facial nerve is also thinned. These maneuvers should be performed under continuous facial nerve monitoring.
Transcochlear Petrosectomy
This technique provides the maximal area of exposure by completely removing the petrous bone. The external auditory canal is transected and oversewn. The initial drilling is as described for the translabyrinthine technique. The facial nerve canal is completely skeletonized and opened to allow the removal of the facial nerve. The greater superficial petrosal nerve (GSPN) can be divided to permit the posterior transposition of the facial nerve. This maneuver moves the facial nerve out of the surgical corridor and allows the complete removal of the internal auditory canal and cochlear apparatus. Alternatively, the GSPN can be preserved and the course of the facial nerve skeletonized. This maneuver preserves the blood supply of the facial nerve and usually does not produce a postoperative facial paralysis. However, the facial nerve then remains directly in the surgeon’s working space throughout the procedure. Drilling then continues until the internal carotid artery has been skeletonized up to the siphon. This technique provides extensive exposure of the petroclival region at the cost of sacrificing hearing and a facial nerve paresis or paralysis if the GSPN is sectioned. This route is also associated with a significant risk of CSF fistula.
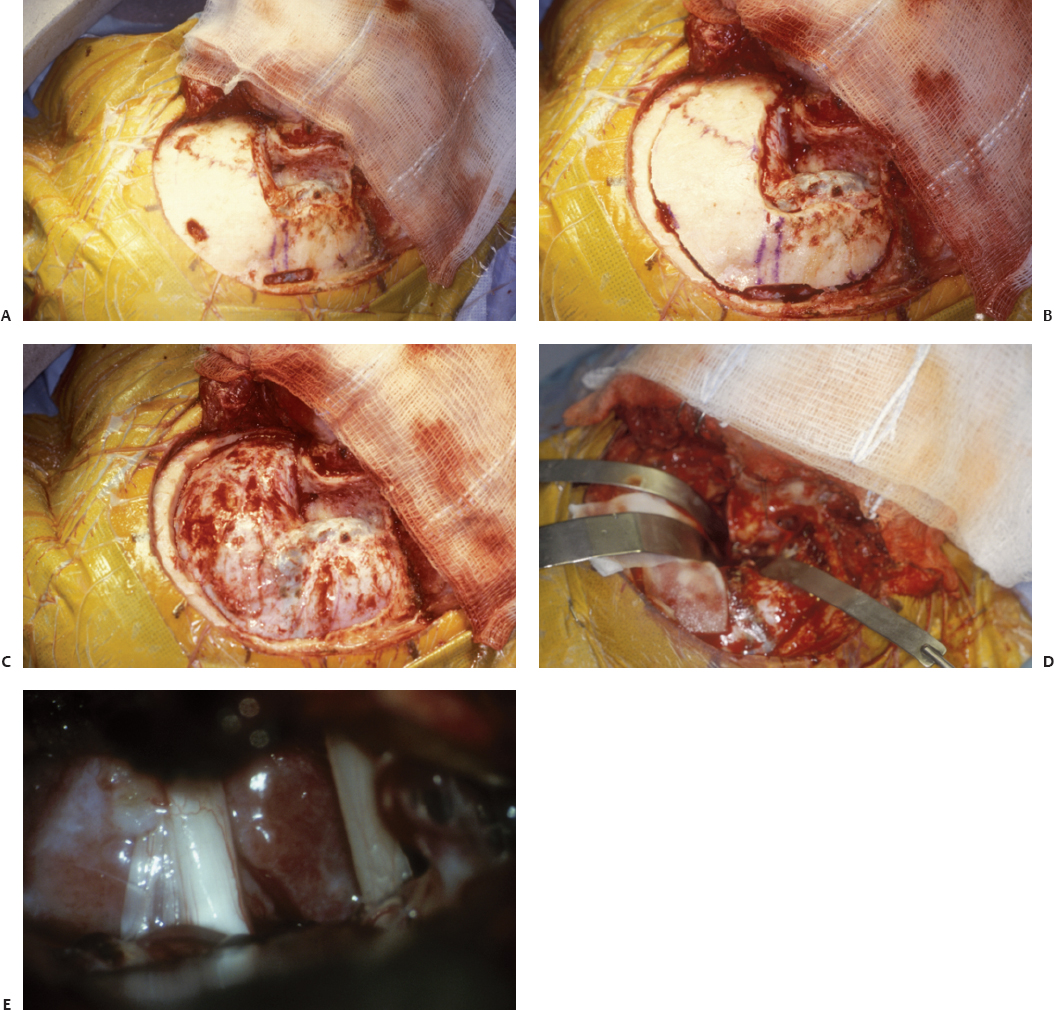
Fig. 12.3 (A) Mastoidectomy and small troughs drilled over the transverse sinus and along the outline of the bone flap. (B) Bone flap turned. (C) Posterior transpetrosal approach bone flap removed. (D) Dural opening. (E) Exposure of a petroclival meningioma medial to cranial nerves V and VIII.
Once the bony exposure is complete, the dura is opened along the inferior edge of the temporal lobe and along the presigmoid sinus dura. The superior petrosal sinus is divided with the bipolar coagulator if it is small and tantalum vascular clips if large. The dural opening then proceeds posteriorly along the superior aspect of the transverse–sigmoid sinus junction, to the point just anterior to the entrance of the vein of Labbé into the dura near the transverse sinus. Great care must be taken to avoid injury to the vein of Labbé. The tentorium is then divided from a lateral to medial direction just behind the posterior edge of the superior petrosal sinus. Temporal bridging veins entering the tentorium should be saved. The medial edge of the tentorium is divided just behind the entrance of cranial nerve IV into the tentorial edge. Once the tentorium is divided, the sigmoid sinus can be displaced posteriorly, and the exposure improves substantially.
After the dura and arachnoid have been opened to permit CSF drainage and relaxation of the brain, the tumor is exposed. The surgeon must orient himself or herself because the normal appearance of the anatomy can be significantly distorted. Tumor removal proceeds according to general neurosurgical principles. Primarily, early devascularization is combined with central debulking. Tumor removal is accomplished by working in the spaces between the cranial nerves. Most petroclival meningiomas receive their vascular supply from the meningohypophyseal trunk off the internal carotid artery. This area of the tumor along the petrous apex should be attacked first. After the initial debulking, dissection proceeds along the tumor capsule. Dissection must remain within the arachnoid planes if the tumor is to be separated successfully from critical vascular and neural structures.
A combination of suction, bipolar coagulation, microscissors, ultrasonic aspiration, and/or CO2 laser are used for the major debulking. During the debulking, the surgeon must maintain a clear mental picture of the expected anatomy and possible location of displaced structures. In particular, the abducens nerve and the basilar artery can be embedded within the tumor and difficult to identify. Tumor debulking proceeds from less critical superficial areas to deeper regions where critical structures may be found.
After generous debulking and relaxation, the tumor capsule is dissected free from the surrounding structures. Meticulous care must be taken to maintain the dissection within the arachnoid planes, however difficult it may be to achieve this goal. Frequently, one must alternate between further debulking and capsule removal. When most of the tumor has been debulked, the thinned-out capsule is carefully dissected free of surrounding structures.
With large extensive tumors, the lower cranial nerves are often involved along the lower pole of the tumor. The facial and vestibulocochlear nerves are usually displaced laterally and inferiorly. Dissection along these nerves can be enhanced with the use of monitoring and a facial nerve stimulator. The abducens nerve is typically within the tumor or stretched anteriorly. It is best to identify this nerve proximally and follow it distally into its dural entrance. Similarly, the trigeminal and trochlear nerves are identified. Occasionally, the dura over Meckel’s cave is opened to remove tumor that has grown into this region.
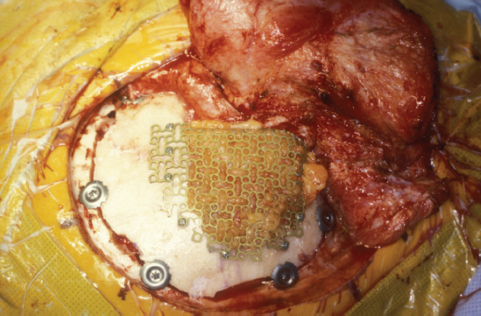
Fig. 12.4 Reconstruction using an abdominal fat graft covered with a titanium mesh. The edges of the mesh must be turned inward or they will protrude through the skin months later as the fat atrophies.
The basilar artery is usually within the tumor or displaced contralaterally. Utmost care must be exercised when dissecting this vessel from the tumor capsule, particularly its critical perforator vessels. It is often wiser to leave a remnant of firmly attached capsule to the basilar artery than to risk neurologic devastation from injuring the perforators or the basilar artery itself.
Closure begins with an attempt to repair the dura in a water tight fashion. If a dural cuff exists deep in the exposure, around the labyrinth, then a free piece of parietal periosteum or temporalis fascia is used to repair the dura primarily. If there is no deep dura to sew to, the temporalis muscle–parietal periosteum flap is rotated down into the mastoid defect and tacked to the dural edges as possible. An additional 2 cm by 2 cm patch of temporalis fascia can be placed over the mastoid antrum, to prevent the temporalis muscle or abdominal fat graft from bulging into the middle ear onto the incus. Some surgeons prefer to use hydroxyapatite bone cement to seal the mastoid antrum. The mastoid defect is then filled with an abdominal fat graft. Titanium mesh can be placed over the fat graft to prevent a sunken mastoid area (Fig. 12.4). Placing mesh over a fat graft in the mastoid must be done with caution. Any sharp edges on the mesh must be bent inward, or over time they will protrude through the scalp as the fat graft atrophies and surrounding skin retracts. There is also anecdotal evidence that mesh, when used over a fat graft in the mastoid, can contribute to late postoperative CSF leak. As the fat graft in the mastoid atrophies, it can adhere to the mesh. This causes the fat to pull away from the presigmoid dura as well as the antrum/middle ear, allowing CSF to leak down the eustachian tube. If mesh is used, the sternocleidomastoid periosteum flap is then sutured back into its normal position over the mesh. The skin is closed in the usual fashion.
♦ Frontotemporal/Orbitozygomatic Craniotomy
Indication
This approach is reserved for petroclival meningiomas with primarily supratentorial extension. Its strength lies with its familiarity to the neurosurgical community, the width of exposure, and the ability to work from multiple angles. The drawbacks of this approach are the potential for injury of the frontal branch of the facial nerve,12 the danger to orbital contents, the potential need for extensive arachnoid dissection with manipulation of the frontal and temporal lobes, the risk of injury to sylvian vessels, and poor access to the middle and lower clivus.
Procedure
The head is rotated from 30 to 60 degrees depending on the location of the lesion. Positioning is such that the malar eminence represents the zenith of the operative field. The incision starts at the inferior border of the zygoma and extends in a gentle curve to the level of the contralateral midpupillary line, terminating behind the hair line. The skin is elevated anteriorly until the superficial temporal fat pad is encountered. Further dissection anteriorly may lead to injury of the frontalis branch of the facial nerve. The temporal fascia is then incised in the line of the skin incision to the level of the superior temporal line. Next the fascia is incised anteriorly approximately 0.5 cm below the superior temporal line, terminating just behind the lateral orbital rim. This leaves a generous cuff of muscle for reattachment of the temporalis muscle at the end of the operation and protects the frontal branch of the facial nerve. The temporal fascia is dissected off the underlying muscle so as to expose the zygoma, the malar eminence, and supraorbital rim. The underlying muscle is then dissected from the cranium and is retracted inferiorly. The periorbita is dissected beginning at the lateral edge of the orbital rim and proceeding medially. At the end of the dissection the entire orbital rim lateral to the supraorbital nerve, the malar eminence, and the zygoma should be completely exposed. An orbitozygomatic (OZ) osteotomy significantly increases the exposure.13,14 It can be performed as one or two pieces.15 In our practice it is completed in two separate steps (Fig. 12.5). First a standard pterional craniotomy is performed. The reciprocating saw is used to perform the OZ osteotomy. Six cuts are required for the removal of the OZ rim.16 The first cut is made at the base of the zygomatic arch. The second cut begins at the inferolateral edge of the zygoma near the malar region and proceeds halfway along the malar eminence to the lateral orbital rim. A third cut begins intraorbitally in the inferior orbital fissure and extends posterolaterally until it meets the edge of the second cut. The fourth cut begins intraorbitally 1 to 2 mm lateral to the supraorbital notch and proceeds posteriorly along the orbital roof approximately 4 cm and medially to the superior orbital fissure. A fifth cut is made from the inferior orbital fissure posteriorly to the temporal region. The final cut is made from the superior orbital fissure inferiorly to meet the previous cut. After the OZ osteotomy is completed, the sphenoid wing is drilled so as to allow for an unobstructed view. The dura is then opened in a standard frontotemporal fashion. After the intradural resection is completed the dura is closed and the bony components replaced and fixed into their anatomic location. The muscle, fascia, and skin are reapproximated in the standard fashion.
♦ Middle Fossa Approach
Indication
This approach is reserved for small lesions that extend into the middle fossa and posterior cavernous sinus, with an inferior extent that is above the internal auditory canal. The main advantage of this approach is that it is fast and is primarily extradural in nature. The drawbacks are the risks of CSF leak, injury to the carotid artery, retraction injury to the temporal lobe, and limited inferior exposure along the clivus.
Procedure
The head is placed so that the zygoma is parallel to the floor. The vertex of the head may be tilted 10 to 20 degrees below the horizontal so that gravity allows the temporal lobe to fall away from the temporal fossa floor. An incision is made from in front of the tragus extending superiorly and slightly posteriorly, until it is above the origin of the temporalis muscle. A temporal craniotomy is turned that is flush with the middle fossa floor (Fig. 12.6). The inferior exposure usually enters the mastoid air cells. Some authors recommend resection of the root of the zygomatic arch including the glenoid fossa, the roof of the external ear, and the superior third of the mastoid process.17,18 The approach is extradural and is assisted greatly by the use of a dedicated middle fossa retractor. There are six key structures that must be identified during this exposure. The first is the posterior petrous ridge. This ridge determines the posterior aspect of the exposure. The second is the arcuate eminence, which overlies the superior semicircular canal. The third is the GSPN, which is left in its place along the middle fossa floor by dissecting the dura off the temporal bone in a posterior to anterior direction, starting along the posterior petrous ridge. Care should be taken to avoid traction on the greater superficial nerve and geniculate ganglion. Next the middle meningeal artery is identified and divided. This maneuver allows additional anterior extradural dissection exposing the mandibular division of the trigeminal nerve as it travels into the foramen ovale. The fifth structure identified is the petrous carotid artery. This is usually found medial and just behind the third division of the trigeminal nerve as it enters the foramen ovale. It is often incompletely covered by dehiscent bone or cartilage. The final structure to be identified is known as the trigeminal impression. This bony depression over the petrous apex is made by the trigeminal nerve as it enters Meckel’s cave, and helps to define the top of the medial petrous apex.
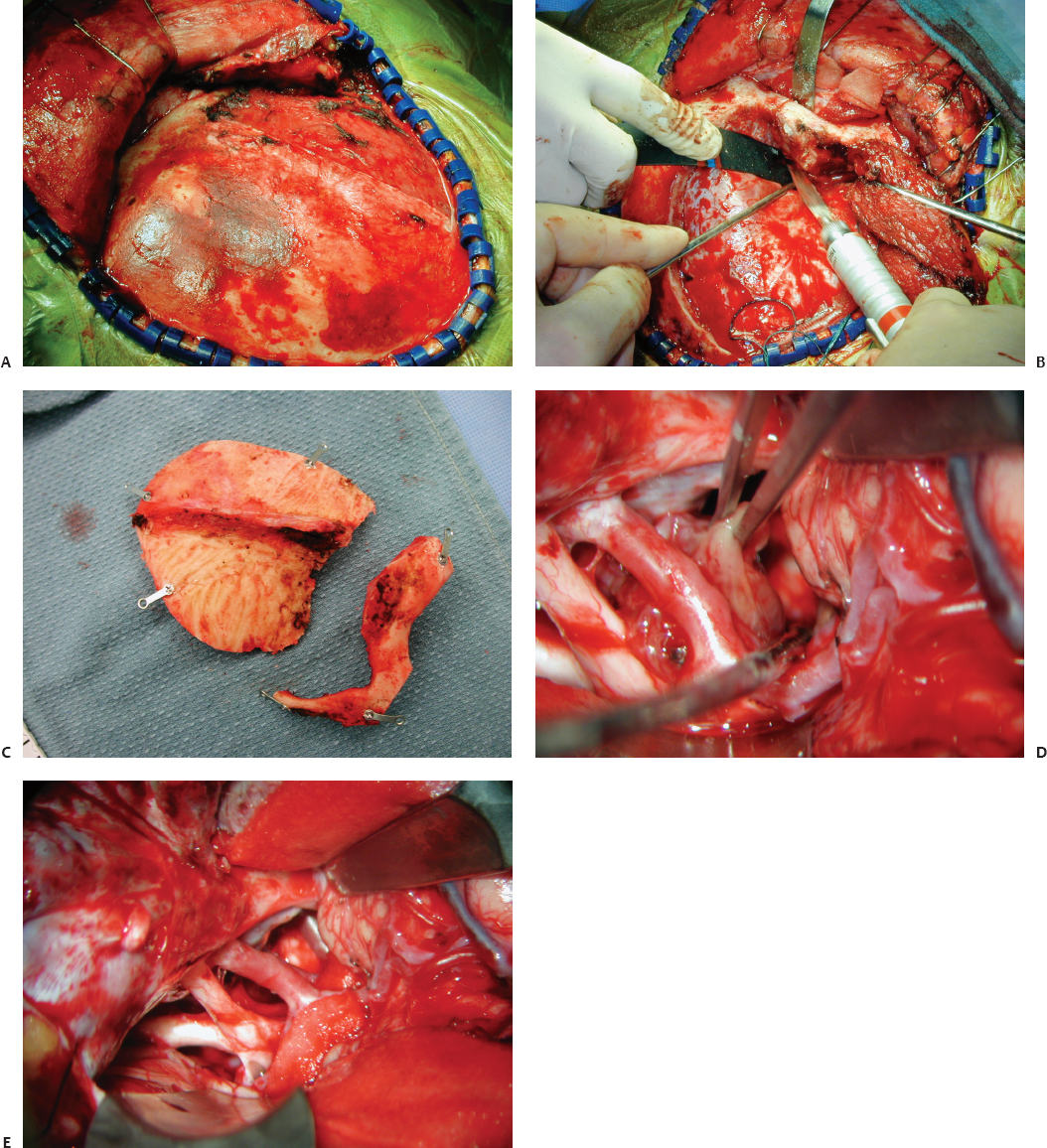
Fig. 12.5 The two-piece orbitozygomatic approach. (A) Scalp incision. (B) Osteotomy cuts. (C) Two-piece orbitozygomatic bone flaps. (D) Exposure of a petroclival meningioma by the orbitozygomatic bone flap. (E) View after resection of a petroclival meningioma.
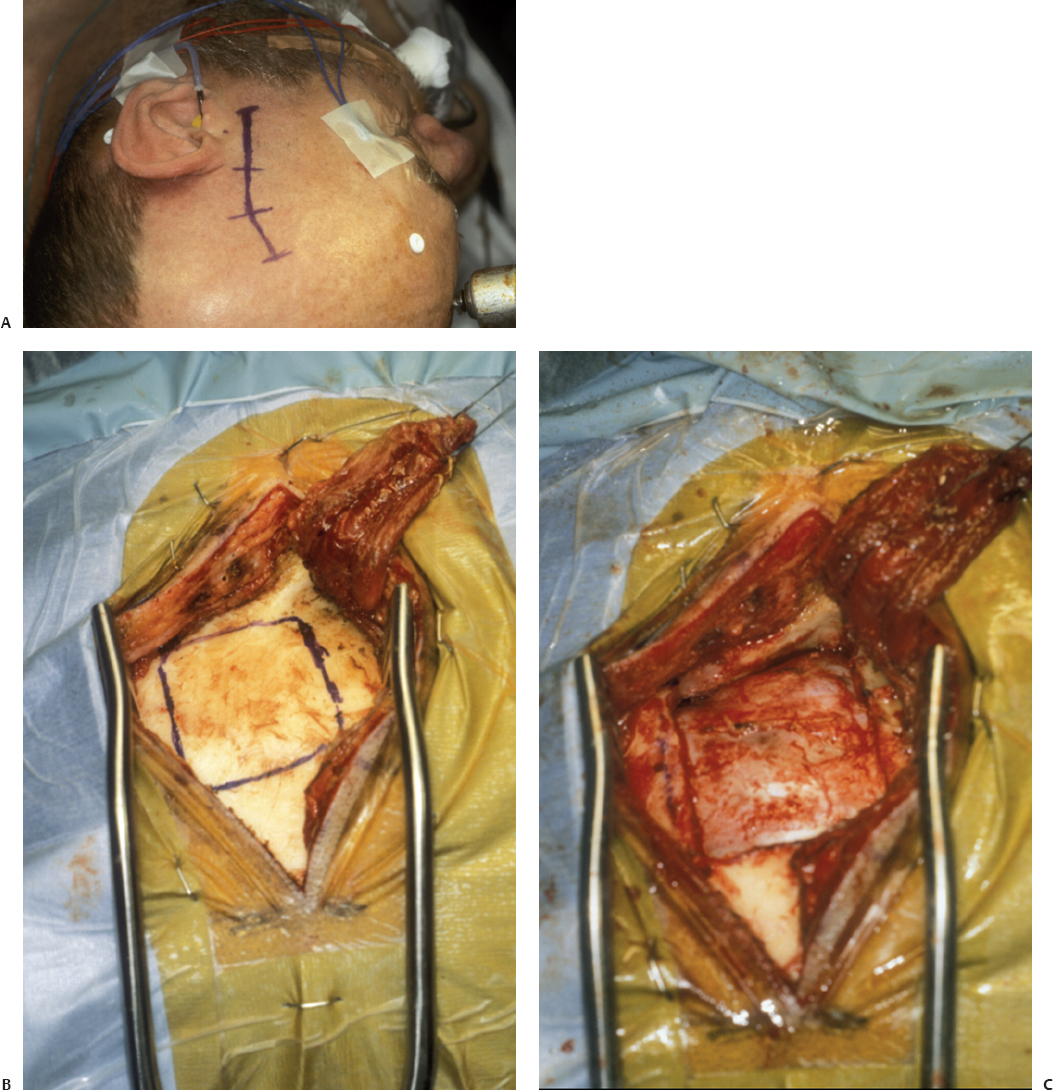
Fig. 12.6 Middle fossa approach. (A) Location of incision. (B) Bone flap outline. (C) Bone flap removed.
All drilling should be performed carefully, usually with a high-speed diamond drill. First the petrous apex is removed exposing the posterior fossa dura. The bone removal proceeds until the internal auditory canal is exposed posteriorly, and the posterior edge of the horizontal segment of the internal carotid artery is exposed anteriorly. Care must be taken to avoid entrance into the cochlea, if this structure is to be preserved. This results in exposure through Kawase’s triangle, which provides access to the superior clivus.19–22 This region is defined anteriorly by the mandibular division of the trigeminal nerve and the internal carotid artery, posteriorly by the internal auditory canal and the superior semicircular canal, laterally by the cochlea and GSPN, and medially by the petrous ridge. The dura along the lateral edge of the third division of the trigeminal nerve is opened and the dura elevated up off of V3. The dura above and below the superior petrosal sinus is opened next. The superior petrosal sinus is ligated and divided. The tumor is then removed, generally by internal debulking, followed by microsurgical capsular dissection. At the end of the intradural resection, the dura, craniotomy, muscle, and skin are closed in the standard fashion. Particular attention must be paid to any exposed air cells. If necessary, a temporalis muscle with attached parietal periosteum can be rotated in under the temporal lobe to seal the mastoid and petrous apex air cells. Meticulous use of bone wax or bone cement may also help minimize the risk of postoperative CSF leak.
♦ Suboccipital Approach
Indication
This approach is reserved primarily for small tumors limited to the infratentorial space that are more petrous than clival in origin. It also can be used in older patients who have some degree of cerebellar atrophy, thus limiting the need for cerebellar retraction. It allows excellent visualization of the petrous bone in a relatively rapid fashion. When modified to a far lateral approach, it extends the operative field to include the lower clivus, including the foramen magnum.23,24 Drawbacks of this approach are a smaller working space, the risk of injury to critical arteriovenous structures and cranial nerves, and CSF leak. The far lateral approach may also result in atlanto-occipital instability if the majority of the occipital condyle is removed.
Procedure
The patient is placed in the lateral decubitus position with a roll under the ipsilateral shoulder. The neck is placed in slight flexion with rotation of the head, away from the surgical site. Care is taken to avoid overrotation and obstruction of the contralateral jugular venous outflow. A linear incision is placed approximately 2 to 3 cm behind the ear. The mastoid emissary vein is occluded with bone wax. A trough of bone is removed along the inferior border of the transverse sinus, as well as along the posterior border of the sigmoid sinus until the mastoid emissary veins entrance into the sigmoid is identified and divided (Fig. 12.7). The dura over the posterior fossa dura is then stripped off the bone with a Penfield 3. A footplate may then be used to complete the craniotomy. All mastoid air cells should be waxed. The dura is then opened along the sigmoid and transverse sinus, leaving a cuff of dura for resuturing at the end of the case. The cerebellar hemisphere is left covered with dura as much as possible.
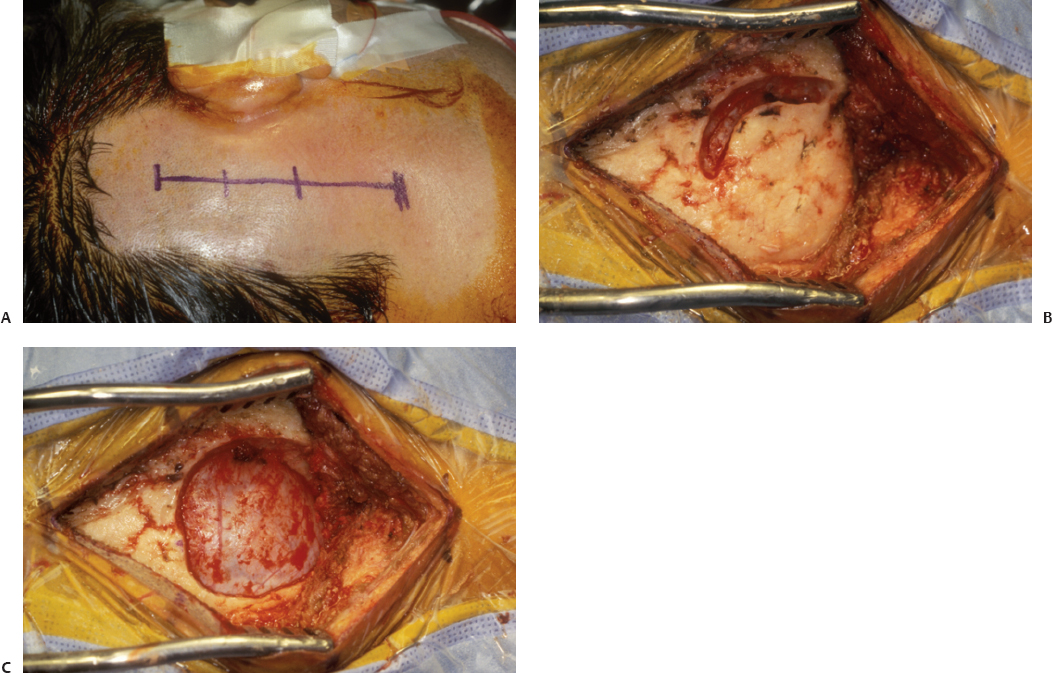
Fig. 12.7 Suboccipital approach. (A) Location of incision. (B) Trough drilled along the inferior border of the transverse sinus and posterior border of the sigmoid sinus. (C) The suboccipital bone flap removed.
The arachnoid over the cisterna magna is opened. This allows for removal of CSF, which provides decompression of the CSF spaces, increases the initial exposure, and limits the need for cerebellar retraction. The cerebellar cortex should be draped with a layer of cottonoid or Telfa. The surgeon can then proceed with arachnoid dissection and removal of the tumor. If intradural drilling is performed, packing of all exposed CSF spaces with Gelfoam and copious irrigation may help reduce the incidence of postoperative headache and nausea.25 Special care should be taken during the closing stages of this procedure. Dural closure should be watertight. Air cells should be meticulously waxed. Fascial and muscle closure should be performed in multiple layers. The skin is closed with a running stitch. All of these maneuvers are intended to minimize the risk of postoperative CSF leak.
Clinical Pearls
1. Turning a combined supra-and infratentorial bone flap without tearing the convexity dura or injuring the underlying venous sinuses can be difficult. Performing the mastoidectomy first aids in the exact identification of the location of the transverse sinus and makes the turning of this bone flap much easier.
2. When performing the posterior transpetrosal approach, elevating both an anterior-based temporalis muscle–parietal periosteum flap and an inferiorly base sternocleidomastoid periosteum flap during the initial exposure is very useful in performing a closure that minimizes the risk of postoperative CSF leak.
3. When performing the posterior transpetrosal approach, the entrance point of the vein of Labbé into the transverse sinus should be determined preoperatively. This can usually be determined from an MRI/magnetic resonance venogram (MRV), but in some cases will require a preoperative arteriogram. The sigmoid sinus should be preserved, especially if the torcula is not patent.
4. When performing the posterior transpetrosal approach, temporal bridging veins entering the tentorium can be preserved by sectioning the tentorium anterior to their entrance into the tentorium and placing the temporal lobe retractor under both the tentorium and the temporal lobe.
5. A cosmetic mastoidectomy can be performed via removal of the outer table of the mastoid bone for later replacement26 or by covering the abdominal fat graft placed in the mastoid defect with titanium mesh. However, if mesh is used, all sharp edges must be turned inward, or they will protrude through the skin months or years later as the fat atrophies.
6. Preoperative embolization of a petroclival tumor can decrease intraoperative blood loss, allow for better visualization, and facilitate radical excision of the tumor while reducing the risk of neurologic morbidity.27,28 However, embolization of the meningohypophyseal trunk is difficult and should be performed only by the most skilled interventionalist. If the patient is not embolized, the area of the tumor closest to the meningohypophyseal trunk should be attacked first to devascularize the tumor as much as possible.
7. Drilling a trough through the skull along the inferior transverse sinus and posterior sigmoid sinus until the entrance of the mastoid emissary vein is ligated decreases the incidence of significant bleeding when turning a suboccipital bone flap with a footplate attachment.
8. Stereotactic radiosurgery allows the surgeon to compromise resection in favor of preservation of function. When critical structures are involved by the tumor that cannot be dissected free, a carpet of tumor may be left on these structures and the remnant tumor irradiated in the postoperative period. Radiosurgery has a very low morbidity rate and enables good tumor control of such remnants.29–36
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








